Dr. Leisha Emens of the University of Pittsburgh Medical Center's Hillman Cancer Center discusses the overall survival in the KATE2 study.

Your AI-Trained Oncology Knowledge Connection!

Dr. Leisha Emens of the University of Pittsburgh Medical Center's Hillman Cancer Center discusses the overall survival in the KATE2 study.

This video reviews the latest trials and research on immunotherapeutic agents being tested in breast cancer.
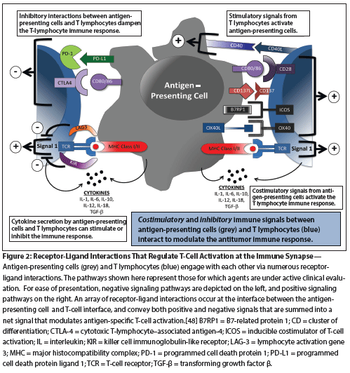
The immune system is active in breast cancer, playing a dual role in tumor progression and in immune surveillance. Infiltrating immune cells are both prognostic and predictive of response to standard breast cancer therapies.
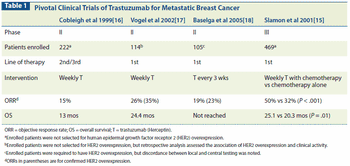
This article reviews clinical data informing the effective management of HER2-positive metastatic breast cancer, including the optimal sequence of HER2-targeted agents.

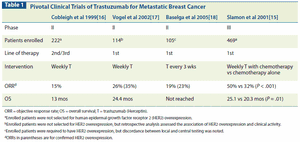
Trastuzumab (Herceptin) is a therapeutic monoclonal antibody specificfor the human epidermal growth factor receptor type 2 (HER2), acell-surface tyrosine kinase receptor overexpressed by 25% to 30% ofbreast cancers. The drug is now regarded as one option for standardtherapy in HER2-overexpressing metastatic breast cancers. It is associatedwith a moderate response rate as a single agent, and in combinationwith standard chemotherapy, can produce greater response ratesand prolong the survival of women with advanced breast cancer. Itsactivity in metastatic breast cancer has led to active clinical trials examiningits potential role in the neoadjuvant and adjuvant settings.The successful clinical development of trastuzumab provides furtherproof of principle that biologically targeted therapies can have a profoundimpact on the management of breast cancer. Here we review theclinical development of this novel agent, emphasizing the potential fortherapeutic synergy when trastuzumab is combined with both standardchemotherapy and innovative molecularly targeted and biologic agents.

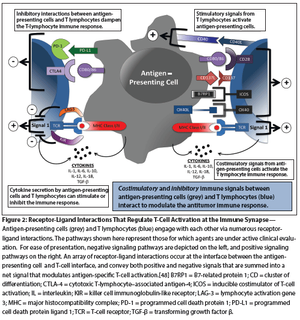
Advances in biotechnology and basic immunology have convergedto create an unprecedented opportunity to use vaccines to harness thepower of the immune system in the fight against breast cancer. Cancervaccines have several therapeutic advantages over more traditionalbreast cancer treatment modalities. First, targeting the antitumorimmune response to critical tumor-specific antigens defines a therapywith exquisite specificity and minimal toxicity. Second, immune-mediatedtumor destruction occurs by mechanisms distinct from those underlyingthe efficacy of chemotherapy and hormone therapy. Thus, immunotherapyoffers an approach to circumventing the intrinsic drugresistance that currently underlies therapeutic failure. Third, thephenomenon of immunologic memory endows immunotherapy withthe potential for creating a durable therapeutic effect that is reactivatedat the onset of disease relapse. Moreover, immunologic memory alsounderlies the potential future use of vaccines for the prevention ofbreast cancer. Early clinical trials have highlighted the promise ofbreast cancer vaccines, and have further defined the challenges facingtranslational scientists and clinical investigators. The judicious applicationof laboratory advances to clinical trial design should facilitatethe development of immunotherapy as an additional major therapeuticmodality for breast cancer, with the potential for breast cancer preventionas well as treatment.

Published: August 1st 2004 | Updated:
Published: March 15th 2013 | Updated:
Published: May 15th 2015 | Updated:

Published: April 10th 2017 | Updated:

Published: September 1st 2003 | Updated:
Published: October 8th 2019 | Updated: