Primary Cutaneous and Systemic Anaplastic Large Cell Lymphoma
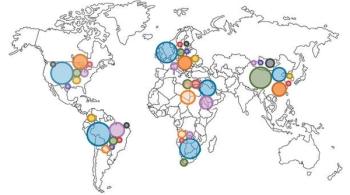
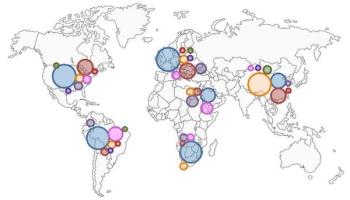
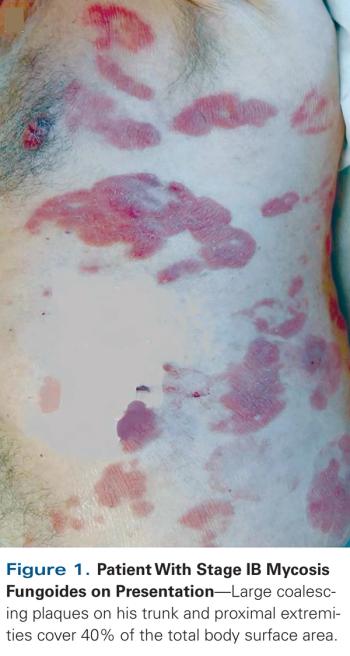
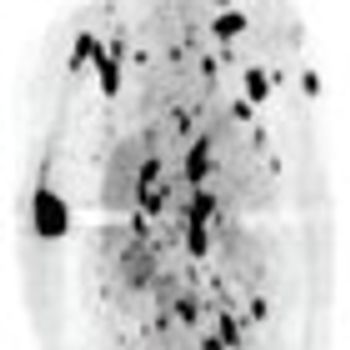
ByChristiane Querfeld, MD, PhD,Irum Khan, MD,Brett Mahon, MD,Beverly P. Nelson, MD,Steven T. Rosen, MD,Andrew M. Evens, DO, MBA, MSc Anaplastic large cell lymphoma (ALCL) is a biologic and clinically heterogenous subtype of T-cell lymphoma. Clinically, ALCL may present as localized (primary) cutaneous disease or widespread systemic disease. These two forms of ALCL are distinct entities with different clinical and biologic features. Both types share similar histology, however, with cohesive sheets of large lymphoid cells expressing the Ki-1 (CD30) molecule. Primary cutaneous ALCL (C-ALCL) is part of the spectrum of CD30+ lymphoproliferative diseases of the skin including lymphomatoid papulosis. Using conservative measures, 5-year disease-free survival rates are>90%. The systemic ALCL type is an aggressive lymphoma that may secondarily involve the skin, in addition to other extranodal sites. Further, systemic ALCL may be divided based on the expression of anaplastic lymphoma kinase (ALK) protein, which is activated most frequently through the nonrandom t(2;5) chromosome translocation, causing the fusion of the nucleophosmin (NPM) gene located at 5q35 to 2p23 encoding the receptor tyrosine kinase ALK. Systemic ALK+ ALCLs have improved prognosis compared with ALK-negative ALCL, although both subtypes warrant treatment with polychemotherapy. Allogeneic and, to a lesser extent, autologous stem cell transplantation play a role in relapsed disease, while the benefit of upfront transplant is not clearly defined. Treatment options for relapsed patients include agents such as pralatrexate (Folotyn) and vinblastine. In addition, a multitude of novel therapeutics are being studied, including anti-CD30 antibodies, histone deacetylase inhibitors, immunomodulatory drugs, proteasome inhibitors, and inhibitors of ALK and its downstream signaling pathways. Continued clinical trial involvement by oncologists and patients is imperative to improve the outcomes for this malignancy.