
AZD1775, a small-molecule, WEE1-targeting TKI, significantly shrunk tumors when administered in combination with cisplatin and docetaxel in a phase I trial.

AZD1775, a small-molecule, WEE1-targeting TKI, significantly shrunk tumors when administered in combination with cisplatin and docetaxel in a phase I trial.

Patients with alcohol-related HCC had worse OS than other HCC patients, mainly due to poorer liver function and more unfavorable tumor characteristics at diagnosis.

Stage III colon cancer patients adhering to ACS Nutrition and Physical Activity Guidelines for Cancer Survivors after diagnosis had a 42% lower risk of death.

Differential methylation in CITED4, as measured in blood, appears to be a promising marker of bladder cancer susceptibility in women.

This slide show reports on a pooled analysis that found up to a fivefold risk of second cancers among smokers, compared with cancer survivors who never smoked.

On November 19, New York City Mayor Michael Bloomberg signed legislation (Tobacco 21 [Intro 250-A]) making New York the first major city to ban the sale of cigarettes, certain tobacco products, and e-cigarettes to anyone under 21 years of age, up from age 18.

At the 2013 ASCO meeting, investigators of a large Danish study have concluded that surveillance alone following surgery for stage I seminoma is sufficient, sparing patients in this setting from the unnecessary expense and associated toxicities of chemotherapy and radiation treatment.

A study from the Mayo Clinic/University of Iowa assessing the use of surveillance CT scans to detect DLBCL relapses found that the majority of relapses occurred outside of regularly scheduled visits and concluded that it is important to educate patients to be more alert to signs and symptoms of relapse.
Results from the German AIO KRK-0306 study, FIRE-3, show that the addition of cetuximab (Erbitux) to chemotherapy rather than bevacizumab (Avastin) increased overall survival by nearly 4 months in patients with KRAS wild-type metastatic colorectal cancer.
Data presented at ASCO from the phase III DECISION trial showed treatment with the TKI sorafenib (Nexavar) delayed disease progression by 5 months in patients with metastatic differentiated thyroid cancer that had progressed on radioactive iodine.
Results from the phase III GOG 240 trial, presented at ASCO, showed the addition of bevacizumab (Avastin) to standard chemotherapy improved overall survival by nearly 4 months in women with advanced or relapsed cervical cancer.

Nurses from a New Jersey cancer center have adapted several key plans from the CDC, ONS, and other societies to create a comprehensive infection-control plan.

Oncology nurses from a community hospital in Cincinnati, Ohio implemented a two-pronged process for the safe handling of chemotherapy, improving nurses’ satisfaction and comfort levels regarding chemotherapy safety.

A team of nurses and physicians from Huntsman Cancer Institute successfully implemented key changes in their Patient Acute Change Team that increased nurse involvement and reduced the number of codes by 90% in the outpatient setting since they were put in place 2 years ago.

The integration of information technology into healthcare-such as EHRs and patient portals-is growing by leaps and bounds, and cancer care is no exception. Judy Murphy from HHS provides important perspectives on this topic.

A holistic nursing interventions study, presented at the ONS Congress, highlights its ability to reduce anxiety, nausea, and pain, and improve select vital signs.

February 4 marks World Cancer Day 2013--this year is devoted to dispelling myths about cancer, such as cancer is a death sentence or a disease of the wealthy.

With access to clinical trials growing in a variety of clinical settings, nurses increasingly need to develop core skills and knowledge to safely, effectively, and appropriately care for enrolled patients.
A US and European team has developed a 3D technique that produces sharper breast images than those available with standard CT scanners, allowing earlier and more accurate detection of breast tumors.

A large study of the impact of patient navigation (PN) on breast cancer management has found that women who receive PN are diagnosed faster than non-navigated women, and the difference is most striking among biopsied women.

The AICR, noted for its investigation of links between lifestyle and cancer, has released a free digital book for cancer patients and survivors, with current, evidence-based advice on managing diet and activity during and after treatment.
Researchers have concluded after a large meta-analysis of trials involving more than 29,000 patients that patients taking currently available biologic therapies for rheumatoid arthritis (RA) are not at increased risk of cancer.

Research report from May 2012 ONS Congress interview and poster discussion with Roxann Scheerens, from the Arthur G. James Cancer Hospital and Richard J. Solove Research Institute, Columbus, Ohio.

Research report from May 2012 ONS Congress interview and poster discussion with Deborah Allen, from Duke University.

We’re now entering the fourth decade of HIV/AIDS awareness. Last year marked the 30th anniversary of the earliest report (on June 5, 1981) of what is now known as AIDS (acquired immune deficiency syndrome).

With National HIV Testing Day recently behind us and the 19th International AIDS conference happening later in July in Washington, DC, I would like to share some recent HIV/AIDS statistical, research, and policy highlights.

Two of every three families in the US will at some time have at least one family member diagnosed with cancer, and family caregivers provide more than 75% of cancer care. Demands on family caregivers are increasing in complexity and duration.

A web-based interactive support system tailored for cancer patients reduced patients’ feelings of depression and improved their sense of well being, compared with being given access to resources that are publically available on the internet, results of a large 1-year randomized controlled trial have shown.

A multicenter telephone-interview study has found that cancer patients often perceive that communication problems with healthcare providers have contributed to a breakdown in their care, but very few formally report their concerns.
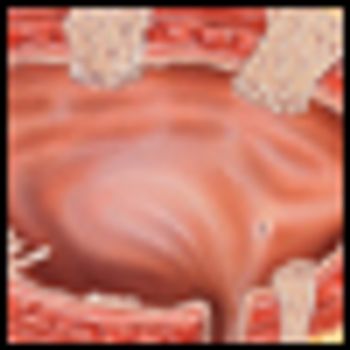
In a multicenter phase III trial of 360 patients with muscle-invasive bladder cancer, synchronous chemoradiotherapy provided better locoregional control without significant added toxicity, investigators for the Bladder Cancer 2001 trial have found.

March 26th 2011
April 21st 2011

June 2nd 2011

December 1st 2007

July 26th 2013