Articles by Jedd D. Wolchok, MD, PhD
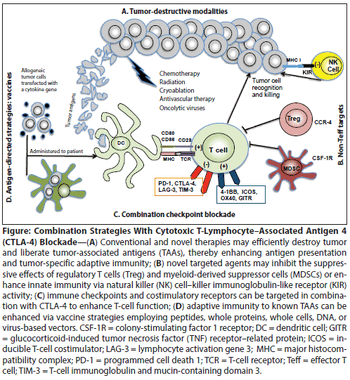
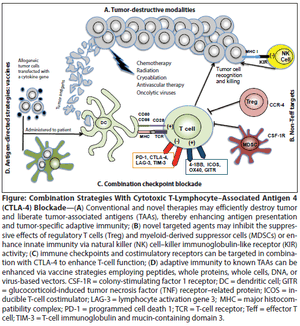
This review will summarize the preclinical and clinical development of CTLA-4–blocking antibodies, discuss recent insights into the biology of CTLA-4 blockade, review the use of these antibodies in combination with established and novel therapeutic modalities, and comment on ongoing questions regarding their administration.

Currently there are only three FDA-approved drugs available for the treatment of metastatic melanoma: dacarbazine, interleukin-2, and the lesser-used hydroxyurea. None of these drugs has been shown to improve overall survival (OS). The review by Thumar and Kluger provides a well-balanced overview of ipilimumab, the first agent to demonstrate a survival benefit in patients with metastatic melanoma.[1] The response to ipilimumab is most notable for its durability, a feature rarely observed in patients with high tumor burden or in response to other systemic therapies. However, a minority of patients (10% to 15%) treated with ipilimumab meet standard criteria for radiographic response. In this commentary, we focus on the question of how we can build on the success of ipilimumab. We briefly review one area of active investigation: the combination of ipilimumab with targeted inhibitors of BRAF.

Surgical resection remains the cornerstone of management for localized renal cell carcinoma. No effective postsurgical adjuvant therapy has been established for