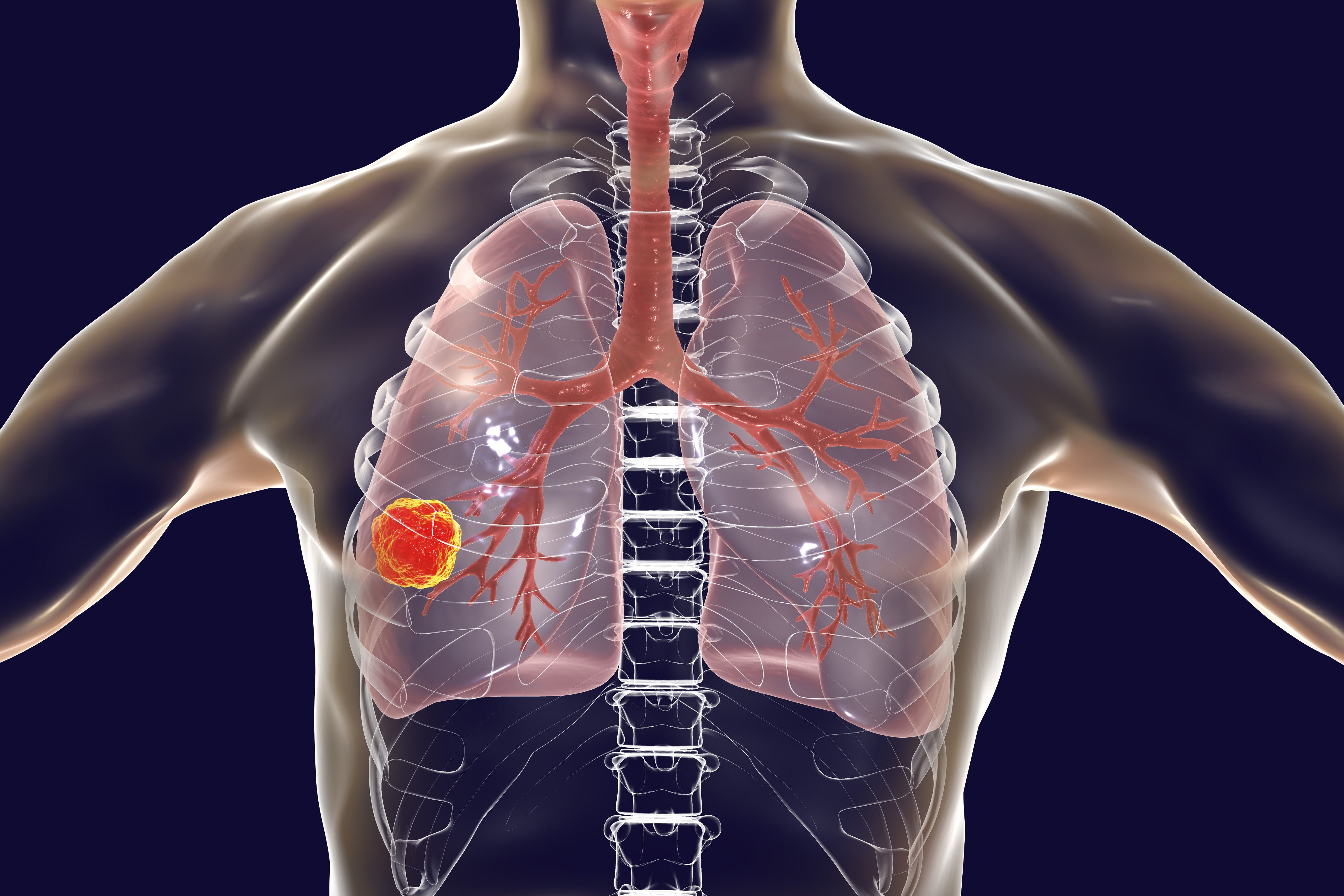
Lung Cancer
Latest News

Latest Videos

More News
The SU2C–LUNGevity Foundation–American Lung Association Lung Cancer Interception Dream Team have developed laboratory models to understand the inflammatory process in the lung’s bronchial cells, and how that relates to early lung cancer development.

The FDA approved a 1500 mg fixed dose of durvalumab (Imfinzi) administered every 4 weeks for the treatment of unresectable stage III non-small cell lung cancer after chemoradiation therapy and previously treated advanced bladder cancer.

The clinical trial is designed to evaluate the efficacy and safety of tislelizumab compared to docetaxel in the second- or third-line setting in patients with locally advanced or metastatic non-small cell lung cancer who have progressed on a prior platinum-based chemotherapy.

Researchers suggested that the tumors of patients with stage I and II early-stage non-small cell lung cancer demonstrate the highest tumor mutational burden and most often display the mutational signature associated with tobacco smoking.
This study found that found that mini-strokes and heart attacks were significantly less common among patients with locally advanced non-small cell lung cancer who underwent proton therapy versus conventional photon-based radiation therapy.

Merck is discontinuing the study after an independent Data Monitoring Committee recommendation, which determined the benefit/risk profile of pembrolizumab in combination with ipilimumab did not support continuing the trial.

The results for the phase 2 SUMMIT trial examining neratinib to treat patients of metastatic non-small cell lung cancer (NSCLC) were recently announced by Puma Biotechnology.

This study indicated that patients with up to 3 lung metastases from primary tumors in other sites who were treated with stereotactic body radiation therapy fared comparably well whether their radiation was delivered in 1 or 4 treatment sessions.

This study found that fragment analysis had the ability to detect large MET exon 14 skipping deletions that were missed by next-generation sequencing in non-small cell lung cancer tumor samples.

The FDA approved expanded claims for the cobas EGFR Mutation Test v2 as a companion diagnostic for a broader group of therapies in the treatment of non-small cell lung cancer.

The FDA granted priority review to the supplemental biologics license application for cemiplimab-rwlc (Libtayo) as a first-line treatment for patients with locally advanced or metastatic non-small cell lung cancer with 50% or more PD-L1 expression.

The FDA accepted and granted priority review to the supplemental new drug application for osimertinib (Tagrisso) for the adjuvant treatment of patients with early-stage EGFR-mutated non-small cell lung cancer after complete tumor resection with curative intent.
Merck announced positive data regarding 2 studies, one examining pembrolizumab in combination with chemotherapy and another in combination with quavonlimab in combination to treat advanced non-small cell lung cancer.

This study found that single dose-per-cycle plinabulin has a similar neutropenia protection benefit as pegfilgrastim among adult patients with non-small cell lung cancer.

Significantly more patients treated with nivolumab (Opdivo) plus chemotherapy before surgery demonstrated no evidence of cancer cells in their resected tissue in the trial compared to those treated with chemotherapy alone.
The study is evaluating sotorasib in 126 patients with KRAS G12C-mutant advanced non-small cell lung cancer who had failed a median of 2 prior lines of anti-cancer therapies.
A study found that the historically higher lung cancer incidence rates for young Blacks compared to Whites in the United States disappeared for men and reversed for women.

A pooled analysis of 3 randomized controlled trials found that pembrolizumab plus chemotherapy demonstrated response and survival improvements with a manageable safety in comparison with chemotherapy alone in PD-L1‒negative advanced non-small cell lung cancer.
The agency approved the first and only immunotherapy treatment as a first-line therapy for adult patients with unresectable malignant pleural mesothelioma.

The findings from this global, open-label, randomized trial supported the FDA approval of atezolizumab for patients with non-small cell lung cancer with high PD-L1 expression, regardless of histologic type, in May.
Post-operative radiotherapy was associated with a nonstatistically significant increase in disease-free survival in patients with completely resected stage IIIAN2 non-small cell lung cancer and therefore cannot be recommended as a standard of care.

Sotorasib showed promising antitumor activity in patients with non-small cell lung cancer that harbor the KRAS p.G12C mutation.

Cemiplimab-rwlc (Libtayo) monotherapy led to a significant improvement in overall survival and progression-free survival in patients with advanced non-small cell lung cancer with PD-L1 expression on at least 50% of their tumor cells.

Based on these data, researchers indicated that adjuvant osimertinib would be an effective and practice-changing treatment in this setting.
Apatinib in combination with gefitinib in the first-line setting demonstrated superior progression-free survival (PFS) in patients with advanced EGFR-mutant non–small cell lung cancer.