Patient-Reported Outcomes of Pain and Related Symptoms in Integrative Oncology Practice and Clinical Research: Evidence and Recommendations
ByW. Iris Zhi, MD, PhD,Danielle Gentile, PhD,Maggie Diller, MD,Anita Y. Kinney, PhD, RN,Ting Bao, MD, DABMA, MS,Viraj A. Master, MD, PhD,Xin Shelley Wang, MD, MPH ABSTRACT
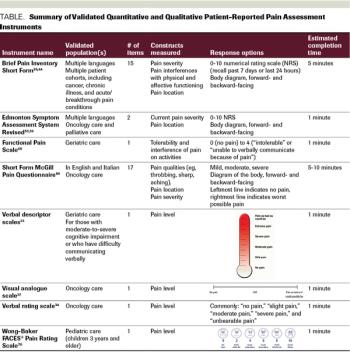
Pain is a primary concern among patients with cancer and cancer survivors. Integrative interventions such as acupuncture, massage, and music therapy are effective nonpharmacologic approaches for cancer pain with low cost and minimal adverse events. Patient-reported outcomes (PROs) that have been validated in many clinical and research settings can be used to evaluate pain intensity, associated symptom burden, and quality of life. Clearly defined, reliable PROs can improve patient satisfaction and symptom control. As integrative oncology continues to evolve and expand, cancer-related pain PROs must be standardized to accurately guide clinicians and researchers. Well-validated pain PROs, such as the Brief Pain Inventory, are among the most commonly used for pain intensity assessment. Multiple symptom assessment tools such as the MD Anderson Symptom Inventory, the Memorial Symptom Assessment Scale, the Edmonton Symptom Assessment System, and the Patient-Reported Outcomes–Common Terminology Criteria for Adverse Events measurement system can also capture pain-associated symptom burden. Electronic PROs provide flexibility in collecting and analyzing PRO data. Clinical trials using carefully selected PROs and rigorous statistical analysis plans are fundamental to conducting high-quality integrative oncology research and promoting utilization of effective integrative interventions to improve patient outcomes. In this review, we aim to summarize current, validated PROs specific to cancer-related pain to aid integrative oncology clinicians and researchers in patient care and in study design and implementation.