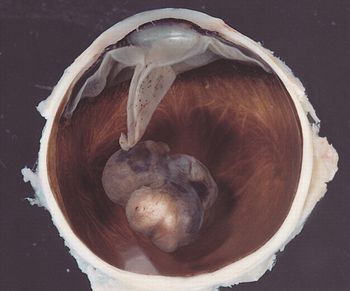
Researchers have discovered that individuals carrying a mutation in the BAP1 gene are at greater risk of developing mesothelioma and uveal melanoma.

Researchers have discovered that individuals carrying a mutation in the BAP1 gene are at greater risk of developing mesothelioma and uveal melanoma.
Scientists at Jennerex, Inc. in San Francisco, and collaborators from University of Pennsylvania and the University of Ottawa in Canada have just engineered a poxvirus, JX-594, to selectively replicate in tumor cells that have an activated EGFR/ Ras pathway, but not in normal tissue.

The FDA has approved crizotinib (Xalkori) for the treatment of late-stage non–small-cell lung cancer patients who express the abnormal anaplastic lymphoma kinase (ALK) gene.

Dr. Ramaswamy Govindan, Washington U at St. Louis, discusses patient screening, new research in targeted therapies, as well as the continued role of traditional chemotherapy in lung cancer.

The NCI has released the results of the almost 10-year National Lung Screening Trial (NLST), which compared two lung cancer-detecting techniques: low dose computed tomography (CT), known as spiral CT, and standard chest X-ray.

A recent NCI study has shown that men have higher cancer mortality rates than do women. The study systematically extracted data from the SEER Database of the NCI, analyzing 29 years of survival statistics data (ending in 2006) from 36 different cancers across the United States.

A study in the AACR journal Cancer Prevention Research, has shown evidence that celecoxib, a cyclooxygenase-2 (COX-2) inhibitor, may be a potent chemopreventive agent for lung cancer.

When I heard about the FDA’s dramatic new step in the anti-smoking fight, I couldn’t help but wonder if it would really make a difference. The new measure requires tobacco companies to add gruesome images to cigarette packages; the images include a corpse, a person’s chest stitched together following heart surgery and even a man with smoke drifting through a hole in this throat.

In this exclusive video, Dr. Harvey Pass discusses surgery in mesothelioma of the pleura and other areas of disagreement.
Results from an ongoing Phase II trial of patients with non-small cell lung cancer (NSCLC) that harbor a specific mutation in the anaplastic lymphoma kinase (ALK) gene reported at ASCO.

Lung cancer remains the leading cause of cancer-related death in the United States. Ongoing research into the molecular basis of lung cancer has yielded insight into various critical pathways that are deregulated in lung tumorigenesis, and in particular key driver mutations integral to cancer cell survival and proliferation.

In 2004, Dr. Thomas Lynch[1] and others[2] first reported the presence of somatic mutations in the epidermal growth factor receptor (EGFR) gene in patients who exhibited great sensitivity to EGFR tyrosine kinase inhibitors (TKIs).

The authors of "ALK-Targeted Therapy for Lung Cancer: Ready for Prime Time," in this issue of ONCOLOGY, address the newest developments in the field of targeted therapies for advanced non–small-cell lung cancer (NSCLC).

Researchers from Memorial Sloan Kettering Cancer Center and the Weill Medical School of Cornell University have shown an association of epidermal growth factor receptor (EGFR) mutations among tumor samples from men and those who smoke cigarettes.
Researchers at UCLA have engineered “vaults,” barrel-shaped nanoscale capsules found in the cytoplasm of mammalian cells, to slowly release chemokine CCL21 into tumors. CCL21 is a protein that, in pre-clinical studies in mice with lung cancer, stimulated the immune system to recognize and attack the cancer cells.

We review the evidence implicating a strong association between chronic inflammation and cancer, with an emphasis on colorectal and lung cancer.
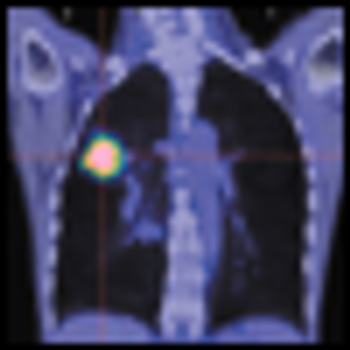
A team of researchers has used mass spectrometry to identify a novel six-biomarker serum test that effectively identified lung cancer in never smokers, and which may have other important diagnostic applications in lung cancer.
Erlotinib (Tarceva) has a low response rate in non–small cell lung cancer (NSCLC) but does improve survival in a subpopulation of patients harboring a wild-type (wt) epidermal growth factor receptor (EGFR) gene.

“Hallmarks of Cancer”, published in the journal Cell in 2000 provided a conceptual framework for the evolution of cancer as well as an all-encompassing review of the cancer field to date. The article is updated in the March 4th, 2011 issue of Cell.

Pfizer’s crizotinib, an oral, selective, small-molecule inhibitor for non-small cell lung cancer (NSCLC) has gone remarkably fast from lab bench to late-stage trials and now to the FDA for review.

“How long have I had this cancer, Doctor?” This is a question that patients frequently ask their oncologist.

In their article, Patrone et al utilize a modified version of Collins’ law to estimate the age of breast, lung, and colorectal cancers. Collins’ law, which states that the period of risk for recurrence of a tumor is equal to the age of the patient at diagnosis plus 9 months, has been applied primarily to pediatric tumors, in particular embryonal tumors.[1,2] The results from the application of Collins’ law to these tumors have been reasonable, although exceptions have been reported and the law is not applicable to all cancers.[3,4] Its utilization in adults in the manner used in this paper is therefore unique.
Exciting advances in understanding the biology of lung cancer have occurred over the last few years.

The authors of “How Long Have I Had My Cancer, Doctor?” have addressed a question often contemplated by patients when they receive a diagnosis of cancer.

The choices that patients and clinicians make when dealing with cancer are dictated by time, whether they are arranging for screening mammography and colonoscopy, compiling treatment plans, or determining follow-up intervals and the age of freedom from follow-up.