Late Effects Clinic: Cardiovascular Late Effects
Chronic or late cardiovascular effects are estimated to occur in less than 5% of patients who receive chemotherapy and mediastinal radiation for all types of cancer.
Chronic or late cardiovascular effects are estimated to occur in less than 5% of patients who receive chemotherapy and mediastinal radiation for all types of cancer.[1-3]
A large number of patients with breast cancer, testicular cancer, Hodgkin's disease, and non- Hodgkin's lymphoma survive the disease; subsequently, these types of cancer tend to be associated with a higher incidence of cardiovascular late effects.
Chronic cardiovascular effects can occur weeks to months or years after cancer treatment. These effects can range from low-voltage QRS complex changes to severe nonreversible cardiomyopathy.
PATIENT OVERVIEW
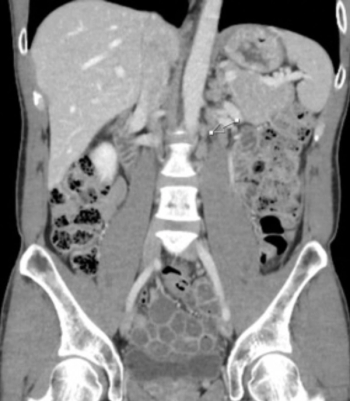
JM is a 36-year-old white male who initially presented to his primary care physician more than a decade earlier, at age 25, with fatigue and a nagging dry cough. At that time, he denied other symptoms such as dyspnea, fever, night sweats, or weight loss.
A chest x-ray was performed, revealing a mediastinal mass. CT (computed tomography) scan of the chest also revealed the mediastinal mass, as well as mediastinal adenopathy. Biopsy of the mass was positive for nodular sclerosing Hodgkin's disease (stage IIa).
As treatment for his Hodgkin disease, JM underwent 12 cycles of chemotherapy with standard dose ABVD: doxorubicin (Adriamycin), bleomycin, vinblastine (Velbane), and dacarbazine (DTIC). After completing chemotherapy, JM went on to receive mantle radiation over a period of 6 weeks.
JM did well for more than 10 years without signs or symptoms suggestive of recurrence until he presented with mild dyspnea, nagging cough, and fatigue. Because his prior treatment for Hodgkin's disease included chemotherapy with an anthracycline, known to pose a risk of cardiotoxic late effects, evaluation included an echocardiogram, which confirmed cardiomyopathy.
JM was treated with digitalis and a vasodilator, and his symptoms improved. The patient continues to be in stable condition and is in remission 1 year after beginning cardiac agents.
NURSING MANAGEMENT
Cardiac functioning should be evaluated during follow-up exams based on the patient's risk factors (see
Methods to evaluate cardiac function include noninvasive monitoring with ECG (electrocardiogram), echocardiography, and radionuclide cardiography. An ejection fraction less than 45% or a decrease in 5% or more from the resting value is considered abnormal. No guidelines exist regarding the optimal interval of cardiac testing or the most effective test to detect cardiac late effects after cancer treatment.
Accurate documentation and monitoring of total cumulative dosages of chemotherapy are essential. Nurses should always encourage smoking cessation, given that smoking increases the risk of late cardiovascular effects. Late onset cardiac toxicity increases with the length of follow-up and occurs in previously treated patients, so emphasizing the need for continued cardiac monitoring in long-term survivors is important.
Cardiac assessments are needed to evaluate patients for signs and symptoms suggestive of cardiac late effects; for instance, a gallop is indicative of cardiac insufficiency. The patient also should be assessed for signs including tachycardia, dyspnea, nonproductive cough, neck vein distention, coarse breath sounds, pedal edema, and fatigue.[3]
Nursing interventions include teaching the patient about energy conservation, managing fluid retention, and minimizing sodium in the diet. Digitalis and ACE (angiotensin converting enzyme) inhibitors can be used as supportive care to enhance cardiac output. Diuretics are used to manage fluid retention. Oxygen and vasodilator medications are often needed to relieve dyspnea. Nurses should teach the patient the importance of routine follow-up for laboratory tests to monitor for electrolyte imbalance.
DISCUSSION
Chronic cardiac toxicities resulting from chemotherapy usually involve nonreversible cardiomyopathy presenting as a classic biventricular congestive heart failure (CHF). Symptoms are classical for CHF, including nonproductive cough, dyspnea, and pedal edema. Unfortunately, patients have a high mortality because of poor response to cardiac medications.[1] Anthracyclines are the most common agents to cause cardiotoxicity through direct damage to the myocyte cells.[2,4] This damage leads to limited contractility causing hypertrophy of the heart muscle. These effects are doserelated, and subsequent cumulative doses considered safe have been established at 550 mg/m2 for doxorubicin, 400 mg/m2 for epirubicin, and 600 mg/m2 for daunomycin.
When mediastinal radiation is to be given, the planned cumulative dose of anthracycline is decreased. Other drugs that can damage myocyte cells include high-dose cyclophosphamide (Cytoxan), fluorouracil (5-FU), paclitaxel (Taxol), ifosfamide (Ifex), and trastuzumab (Herceptin).[1,4,5] Cardiac toxicities can occur in patients treated with radiation therapy where large portals include the heart, most often in patients treated for non-Hodgkin's lymphoma, Hodgkin's disease, and left-sided breast cancer.
The American Society of Clinical Oncology (ASCO), in a clinical evidence review of cardiac (and pulmonary) late effects in adult cancer survivors concluded that, while current incidence may be lower owing to modern radiation techniques, the estimated aggregate incidence of radiation-induced cardiac disease is 10% to 30% by 5 to 10 years post-treatment.[1]
Damage to the cardiac tissue from radiation can continue for months to years after completion of treatment.[6,7] Radiationassociated pericarditis is the most common cardiovascular long-term effect. Coronary artery disease can also occur as a result of radiation causing a direct vascular injury that produces a thickening of the arterial wall.
Cancer survivors who have received cardiotoxic chemotherapy and mediastinal radiation have an increased risk of developing cardiovascular late effects. Limitations on activities of daily living that the individual will experience depend upon the degree of cardiac injury that occurs from cancer treatment. Few patients are prepared for this debilitating effect. Nurses are in the best position to initiate interventions that will assist patients and their families in coping.
Disclosures:
The author has no significant financial interest or other relationship with the manufacturers of any products or providers of any service mentioned in this article.
References:
References
1.
Carver JR, Shapiro CL, Ng A, et al: American Society of Clinical Oncology Clinical Evidence Review on the ongoing care of adult cancer survivors: Cardiac and pulmonary late effects. J Clin Oncol 25(25):3991â4008, 2007. [Epub Jun 18, 2007.]
2.
Keefe DL: Anthracycline-induced cardiomyopathy. Semin Oncol 28(4; suppl 12):2â7, 2001.
3.
Loerzel VW, Dow KH: Cardiac toxicity related to cancer treatment. Clin J Oncol Nurs 7(5):557â562, 2003.
4.
Jones RL, Swanton C, Ewer MS: Anthracycline cardiotoxicity. Expert Opin Drug Saf 5(6):791â809, 2006.
5.
Tsavaris N, Kosmas C, Vadiaka M, et al: Cardiotoxicity following different doses and schedules for 5-fluorouracil administration for malignancy: A survey of 427 patients. Med Sci Monit 8(6): PI51â57, 2002.
6.
Anscher MS, Marks LB, Shafman TD, et al: Risk of long-term complications after TFG-beta 1 guided very high dose thoracic radiotherapy. Int J Radiat Oncol Biol Phys 56(4):988â995, 2003.
7.
Theodoulou M, Seidman AD: Cardiac effects of adjuvant therapy for early breast cancer. Semin Oncol 30(6): 730â739, 2003.
Newsletter
Stay up to date on recent advances in the multidisciplinary approach to cancer.