Recurrent EGFR-Mutated Non–Small Cell Lung Cancer Discovered by Abnormal Mammogram: Adjuvant/Frontline Metastatic Management Options
ABSTRACT: Breast metastasis from extramammary malignancy is rare, with a reported incidence rate of 0.4% to 1.3% in the published literature. The primary malignancies that most commonly metastasize to the breast are leukemia, lymphoma, and malignant melanoma. Here, we report a very rare case of metastatic EGFR-mutated non–small cell lung cancer (NSCLC) in the breast detected by screening mammography. The patient had initially been diagnosed with a clinical stage IIIA NSCLC and had been treated with neoadjuvant chemoradiation followed by curative-intent surgery. Several interesting aspects of the case, along with a discussion of evolving adjuvant and frontline metastatic management options in EGFR-mutated NSCLC, will be presented.
Copur is a medical oncologist/hematologist at Morrison Cancer Center, Mary Lanning Healthcare in Hastings, Nebraska, and is a professor at the University of Nebraska Medical Center. He is also an Editorial Advisory Board member at ONCOLOGY®.

Horn is a pathologist at Mary Lanning Healthcare in Hastings, Nebraska.

Lackner is currently the vice chief of Cardiothoracic Surgery and chief of the section of Thoracic Surgery. He is also the director of the Comprehensive Thoracic Oncology Program at the University of Nebraska Medical Center.

Rodriguez is a board certified diagnostic radiologist at Mary Lanning Healthcare in Hastings, Nebraska and vice president of Hastings Radiology Associates.

Zusag is a radiation oncologist at Morrison Cancer Center, Mary Lanning Healthcare in Hastings, Nebraska.

Introduction
Lung cancer is the leading cause of cancer mortality in both men and women, and the second most common cancer in both genders.1 Non–small cell lung cancer (NSCLC) accounts for 80% to 90 % of all lung cancer cases, half of which present with adenocarcinoma histology.2 In the past decade, several predictive and prognostic biomarkers have become available, significantly changing the treatment landscape in this disease. Current guidelines recommend testing for certain molecular and immune biomarkers in the metastatic setting to assess eligibility for targeted or immunotherapy options.3-5 EGFR driver mutations are the most common predictive biomarkers. Among others with an FDA approved targeted therapy are ALK fusion oncogene, ROS1 gene fusions, BRAF V600E point mutations, NTRK gene fusions, METex14 skipping mutations, RET rearrangements, and PD-L1 expression. Somatic activating mutations in the tyrosine kinase (TK) domain of EGFR behave as oncogenic drivers leading to ligand independent activation of downstream receptor signaling, favoring cell proliferation, survival, and migration.6
The most common EGFR gene mutations in patients with NSCLC are deletions in exon 19 (exon 19del) and a point mutation in exon 21 (L858R), accounting for 45% and 40% of all patients, respectively. Both mutations result in activation of the TK domain, and both are associated with sensitivity to the small molecule EGFR TK inhibitors (TKIs), such as erlotinib, gefitinib, afatinib, osimertinib, and dacomitinib.7 These drug-sensitive EGFR mutations have been named as sensitizing mutations. Other less common mutations (5%-10%) that are also sensitive to EGFR TKIs include exon 19 insertions, p.L861Q, p.G719X, and p.S768I.8 Overall, these sensitizing EGFR mutations are found in 10% of Caucasian patients and up to 50% of Asian patients with NSCLC. 9
Here, we review the rare occurrence of breast metastases from EGFR-mutated NSCLC and discuss the recent developments in the adjuvant and metastatic management of this disease.
Case
A 68-year-old Caucasian woman, lifelong nonsmoker, presented with recurring cough and upper respiratory infection symptoms. Despite initial improvement with antibiotics at each episode, the recurrent nature of her symptoms led to further work-up. A CT scan of her chest revealed a left apical pulmonary mass measuring up to 2.6 x 3.1 cm and a left aorticopulmonary window mediastinal lymph node measuring 2.7 x 3.0 cm (Figure 1). CT-guided biopsy of the lung lesion revealed an adenocarcinoma consistent with lung primary. Further work up with a PET-CT scan confirmed the left apex lung mass with a maximum standardized uptake value (SUV) of 6.4 and the left aorticopulmonary window lymph node with a maximum SUV of 6.7. There were no other hypermetabolic areas. She was staged as cT2cN2cM0, stage IIIA.
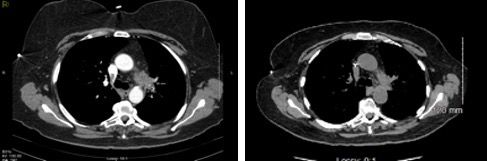
Preoperative chemoradiation (pemetrexed 500 mg/m2 plus cisplatin 75 mg/m2 with concurrent radiation [45 Gy in 180 cGy daily fractions]) led to a decrease in the left apex mass down to 1.7 x 1.4 cm, and only minimal soft tissue fullness remained in the left aorticopulmonary window lesion (Figure 2). She underwent a left video-assisted thoracoscopic surgery with an upper lobectomy and mediastinal lymph node sampling. Pathology revealed left upper lobe 1.6-cm mass consisting of fibroelastosis chronic inflammation macrophage infiltration and reactive epithelial changes consistent with pulmonary tissue with therapy effect, but no residual viable tumor. Similar changes with areas of dense fibrosis and chronic hemorrhage were identified in the level 5 mediastinal node specimen, but no nodal malignancy involvement, confirming complete pathologic response (pyT0pyN0).
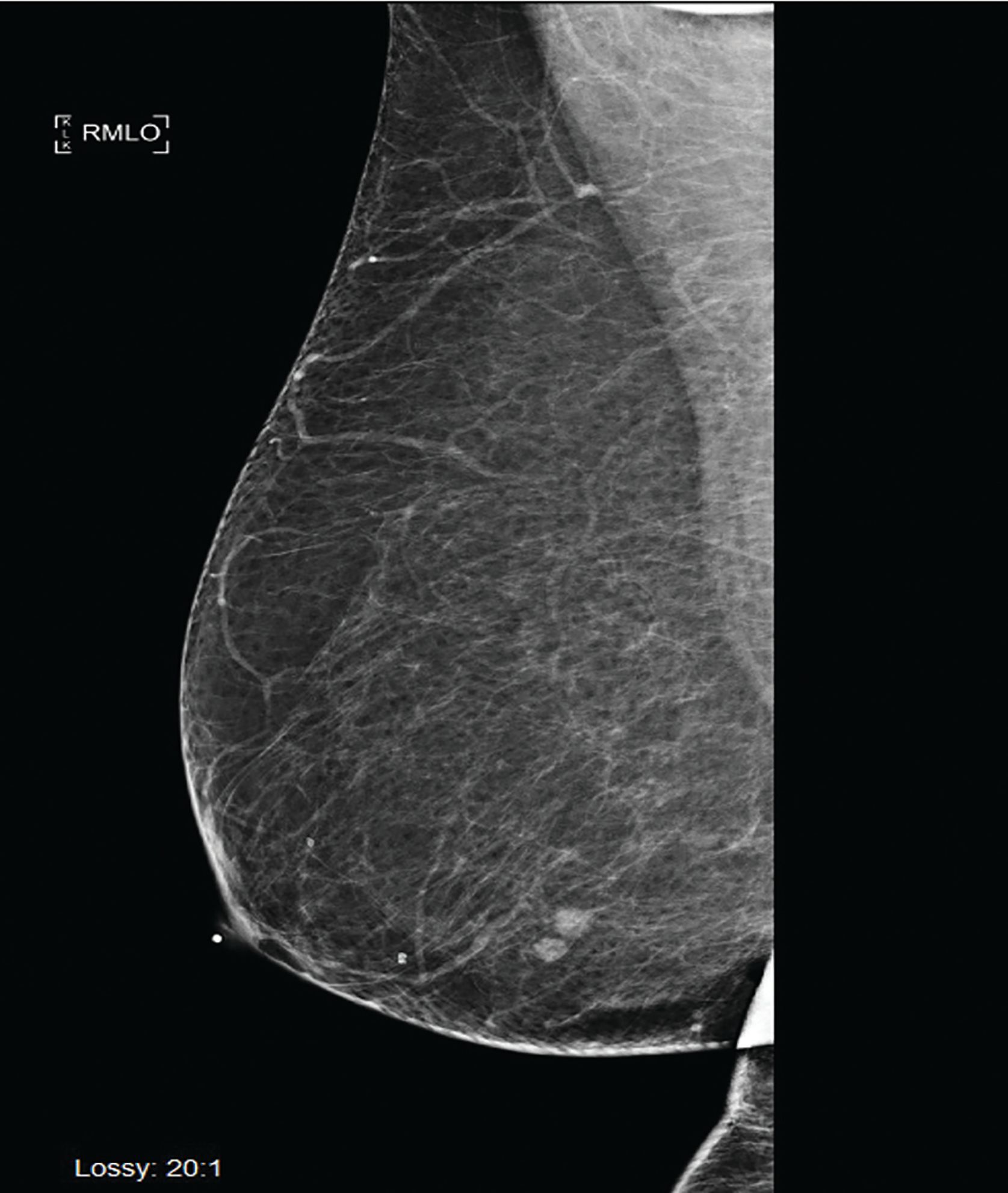
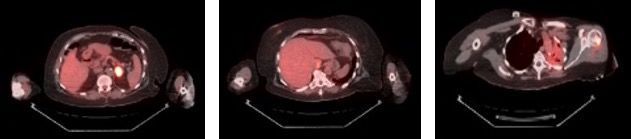
Six months following her surgery, a routine annual screening mammogram revealed 2 new adjacent nodules in the lower central right breast suspicious for breast cancer (Figure 3). Ultrasound guided biopsy of these lesions revealed adenocarcinoma consistent with metastasis from lung primary (Figure 4). A restaging PET-CT scan revealed new hypermetabolic lesions involving caudate lobe of liver, left adrenal gland, and bone lesions in left humerus, right scapula, and right ischium, which were totally asymptomatic (Figure 5).
Discussion
This case illustrates not only a very unusual initial metastatic site for a recurrent EGFR-mutated NSCLC, but it also stimulates a discussion on several evolving management aspects in the adjuvant and metastatic setting of EGFR-mutated NSCLC.
The most common sites of metastasis from NSCLC are liver (33%-40%), adrenal glands (18%-38%), brain (15%-43%), bone (19%-33%), kidney (16%-23%), and abdominal lymph nodes (29%).10 However, an autopsy series has demonstrated that NSCLC may spread virtually to any organ.11 The mammary gland is a rare site of involvement by metastatic disease from extramammary malignancies, likely due to its large areas of fibrous tissue with a relatively poor blood supply. The incidence of breast metastases from extramammary malignancies has been reported to be around 0.4% to 1.3% with a wide range of primary malignant tumors including hematological malignancies, malignant melanoma, lung cancer, renal cancer, ovarian cancer, thyroid carcinomas, and small bowel carcinoids.12-15
Figure 1. CT scan through the lung apices shows a 3.1cm spiculated mass with central low density consistent with necrosis. At the level of the left upper aorticopulmonary window region, there is a 3cm nodal soft tissue mass with central density consistent with necrosis.
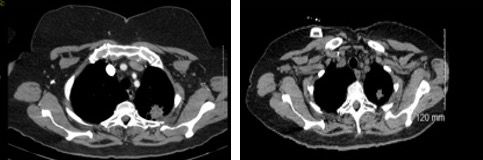
Figure 2. CT scan after treatment shows significant reduction of the left apical pulmonary mass and the left upper hilar nodal mass, consistent with a positive response to chemoradiation.
Among the extramammary malignancies metastasizing to the breast, NSCLC is very rare, with a total of 12 cases reported between 1992 and 2012 in the PubMed database.13,16-24 In a
further updated systematic review of the literature, Mirrielees et al25 identified 31 case reports of NSCLC and 8 cases of small cell lung cancer (SCLC) metastatic to the breast.
Figure 3. Right mediolateral oblique digital mammogram view shows 2 adjacent nodules in the lower breast. Margins are slightly lobulated. The finding was new from the prior mammogram and suspicious for malignancy.
Although breast metastases from primary lung cancer are rare, EGFR-mutated lung cancers metastasizing to the breast are even rarer, with a total of 5 cases reported to date. Fukumoto et al26 reported a case of late breast metastasis from a previously resected EGFR-mutated lung adenocarcinoma. Sato et al27 reported the case of a woman who was initially treated with first-line gefitinib for an EGFR-mutated lung adenocarcinoma. After 12 months of gefitinib, she developed breast metastasis. The metastatic tumor tissue tested positive for T790M mutation. Dansin et al28 reported a case of a woman, aged 52 years, with a breast mass initially diagnosed as a primary triple negative breast cancer; this later proved to be a metastasis from an EGFR-mutated lung adenocarcinoma. Ota et al29 reported a case of metastasis to the breast from a primary lung adenocarcinoma with an L858R mutation in exon 21. Finally, a case of SCLC metastatic to the breast, which transformed from NSCLC during EGFR-TKI maintenance therapy, was reported.30 Our case is thus the sixth reported of EGFR-mutated NSCLC metastatic to the breast.
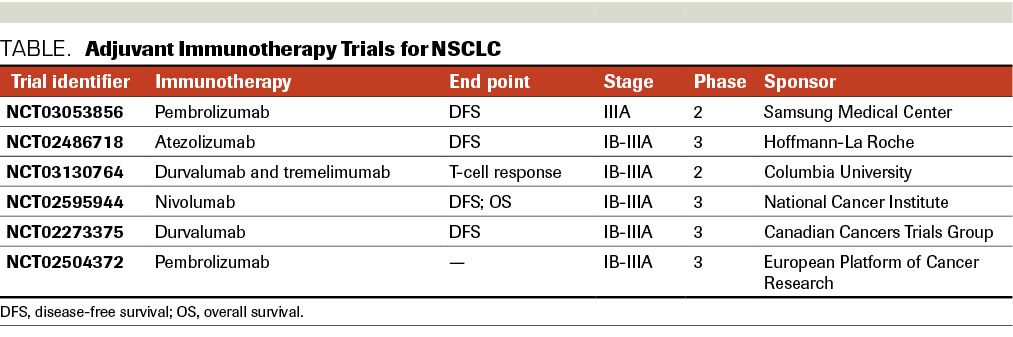
Initial clinical presentation of our case was clinical stage IIIA. The optimal management of patients with potentially operable stage IIIA NSCLC remains controversial. Two randomized trials evaluated the role of surgery in this setting, neither showing an overall survival (OS) benefit.31-33 This is most likely due to the heterogeneity of the patient population with N2 disease in these trials. Similarly, there is no consensus on the role of neoadjuvant chemoradiotherapy vs neoadjuvant chemotherapy alone. OS appears to be similar as long as radiation therapy is given postoperatively, if not given preoperatively.34,35 However, neoadjuvant chemoradiotherapy has been associated with higher rates of pathologic complete response rates and negative mediastinal lymph nodes.36 For patients with unresectable stage III NSCLC, the recommendation is definitive concurrent chemoradiation followed by immunotherapy with durvalumab as consolidation therapy, based on the data from a phase 3 trial.37 On the contrary, there are no established data for the use of adjuvant durvalumab or any other immunotherapy options in resected stage IIIA NSCLC patients. Several ongoing clinical trials are currently testing this concept (Table).37,38 Our patient was treated with neoadjuvant chemoradiation followed by surgery. Despite a complete pathologic response, the unfortunate manifestation of recurrent disease raises the question of possible benefit from adjuvant immunotherapy in this setting.
Figure 4. Breast biopsy, invasive adenocarcinoma (hematoxylin and eosin stain). (A); Breast biopsy, invasive adenocarcinoma. Napsin A immunohistochemical stain (cytoplasmic positivity). (B); Breast biopsy, invasive adenocarcinoma. TTF-1 immunohistochemical stain (nuclear positivity). (C); Breast biopsy, Invasive adenocarcinoma. GATA3 immunohistochemical stain (negative). (D).
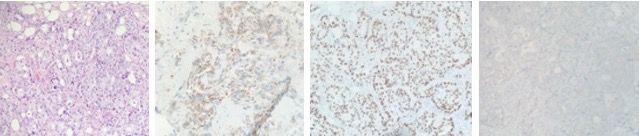
Figure 5. New PET-CT scan. New positive areas of F-18 fluorodeoxyglucose uptake in the left adrenal gland, caudate lobe of the liver, and left humerus. Vague, minimal uptake in the left lung was thought to be pneumonitis.
Once recurrent metastatic disease was established in the patient in our case, molecular and immune biomarker testing was performed, which revealed the EGFR mutation (exon 19del). This finding raises 3 other important management questions: the role of testing for molecular and immune biomarkers of tumor tissue in the adjuvant setting; the role of adjuvant TKI treatment in resected EGFR-mutated NSCLC; and the optimal first-line targeted treatment for metastatic EGFR-mutated NSCLC.
Earlier trials have not demonstrated any benefit of TKI use in the adjuvant setting. However, recent randomized controlled trial results have shown a disease-free survival (DFS) advantage. In a systematic review and meta-analysis of 6 trials incorporating 1860 patients, adjuvant TKIs decreased the risk of disease recurrence by 48%; they improved 2-year DFS but not OS. In subgroup analyses, the DFS benefit was more pronounced in the trials using TKIs rather than chemotherapy, as opposed to trials using TKIs sequentially after chemotherapy.39 In another meta-analysis involving 1960 patients who participated in 5 studies, adjuvant EGFR-TKI treatment was associated with an absolute DFS benefit of 3.1% at 3 years. Finally, in an additional study, adjuvant EGFR-TKI treatment was associated with a marginally statistically significant benefit in OS.40
TABLE. Adjuvant Immunotherapy Trials for NSCLC
Most recently, the first interim analysis from the phase 3 ADAURA trial showed 83% improved overall DFS vs placebo (<.0001). In stage II to IIIA NSCLC patients treated with osimertinib vs placebo, the 2-year DFS was 90% vs 44%, respectively.41 Osimertinib is currently approved for first-line treatment of EGFR-mutated advanced NSCLC and for second-line treatment of EGFR-T790M–mutated advanced NSCLC. The role of TKIs in the adjuvant treatment of EGFR-mutated NSCLC remains hotly debated. Long-term survival data on this topic are eagerly waited.
The optimal first-line treatment regimen for patients with advanced metastatic EGFR-mutated NSCLC is also not well established. A meta-analysis of 25 randomized clinical trials with a total of 5005 patients showed that either the third-generation TKI osimertinib, or first-generation TKIs in combination with chemotherapy, were more effective than first-generation TKIs alone in terms of progression-free survival (PFS) and OS. Second-generation TKIs show significant OS advantage over first-generation TKIs. Based on PFS and OS, osimertinib has the highest probability of being the most effective treatment.42
Finally, the recently published results of the phase 3 RELAY trial, which compared ramucirumab plus erlotinib against erlotinib alone, showed superior median PFS for the combination (19.4 months) vs erlotinib alone (12.4 months).43 While the OS data will not be mature until 2023, statistically projected and calculated OS data were reported to be 58 months for ramucirumab/erlotinib vs 52 months for erlotinib alone.
Outcome
After a discussion of different treatment options, our patient opted for treatment with the ramucirumab/erlotinib combination. Currently, she is receiving her second cycle and tolerating treatments well. A restaging scan is planned in the near future.
References
- American Cancer Society Cancer Facts & Figures: 2020. American Cancer Society. 2020. Accessed September 1, 2020. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2020/cancer-facts-and-figures-2020.pdf
- Li T, Kung HJ, Mack PC, Gandara DR. Genotyping and genomic profiling of non-small-cell lung cancer: implications for current and future therapies. J Clin Oncol. 2013;31(8):1039-1049. doi:10.1200/JCO.2012.45.3753
- Ramalingam SS, Vansteenkiste J, Planchard D, et al; FLAURA Investigators. Overall survival with osimertinib in untreated, EGFR-mutated advanced NSCLC. N Engl J Med. 2020;382(1):41-50. doi:10.1056/NEJMoa1913662
- Garon EB, Hellmann MD, Rizvi NA, et al. Five-year overall survival for patients with advanced non‒small-cell lung cancer treated with pembrolizumab: results from the phase I KEYNOTE-001 study. J Clin Oncol. 2019;37(28):2518-2527. doi:10.1200/JCO.19.00934
- Lin JJ, Cardarella S, Lydon CA, et al. Five-year survival in EGFR-mutant metastatic lung adenocarcinoma treated with EGFR-TKIs. J Thorac Oncol. 2016;11(4):556-565. doi:10.1016/j.jtho.2015.12.103
- Tan DSW, Yom SS, Tsao MS, et al. The International Association for the Study of Lung Cancer consensus statement on optimizing management of EGFR mutation-positive non-small cell lung cancer: status in 2016. J Thorac Oncol. 2016;11(7):946-963. doi:10.1016/j.jtho.2016.05.008
- Langer CJ. Epidermal growth factor receptor inhibition in mutation-positive non-small-cell lung cancer: is afatinib better or simply newer? J Clin Oncol. 2013;31(27):3303-3306. doi:10.1200/JCO.2013.49.8782
- Riely GJ, Politi KA, Miller VA, Pao W. Update on epidermal growth factor receptor mutations in non-small cell lung cancer. Clin Cancer Res. 2006;12(24):7232-7241. doi:10.1158/1078-0432.CCR-06-0658
- O’Kane GM, Bradbury PA, Feld R, et al. Uncommon EGFR mutations in advanced non-small cell lung cancer. Lung Cancer. 2017;109(x):137-144. doi:10.1016/j.lungcan.2017.04.016
- Quint LE, Tummala S, Brisson LJ, et al. Distribution of distant metastases from newly diagnosed non-small cell lung cancer. Ann Thorac Surg. 1996;62(1):246-250. doi:10.1016/0003-4975(96)00220-2
- Matthews MJ. Problems in morphology and behaviour of bronchopulmonary malignant disease. In: Israel L, Chahanian P, eds. Lung Cancer: Natural History, Prognosis and Therapy. Academic Press; 1976:23-62.
- Vizcaíno I, Torregrosa A, Higueras V, et al. Metastasis to the breast from extramammary malignancies: a report of four cases and a review of literature. Eur Radiol. 2001;11(9);1659-1665. doi:10.1007/s003300000807
- Klingen TA, Klaasen H, Aas H, et al. Secondary breast cancer: a 5-year population-based study with review of the literature. APMIS. 2009;117(10):762-767. doi:10.1111/j.1600-0463.2009.02529.x
- Williams SA, Ehlers RA 2nd, Hunt KK, et al. Metastases to the breast from nonbreast solid neoplasms: presentation and determinants of survival. Cancer. 2007;110(4):731-737. doi:10. 10.1002/cncr.22835
- Noguera JJ, Martínez-Miravete P, Idoate F, et al. Metastases to the breast: a review of 33 cases. Australas Radiol. 2007;51(2):133-138. doi:10.1111/j.1440-1673.2007.01681.x
- Masmoudi A, Mathieu M-C, Soria J-C. Breast metastasis from lung adenocarcinoma: a case report. Anticancer Res. 2003;23(2C):1825-1826.
- Ramar K, Pervez H, Potti A, Mehdi S. Breast metastasis from non-small-cell lung carcinoma. Med Oncol. 2003;20(2);181-184. doi:10.1385/MO:20:2:181
- Gómez-Caro A, Pinero A, Roca MJ, et al. Surgical treatment of solitary metastasis in the male breast from non-small cell lung cancer. Breast J. 2006;12(4):366-367. doi:10.1111/j.1075-122X.2006.00278.x
- Yeh C-N, Lin C-H, Chen M-F. Clinical and ultrasonographic characteristics of breast metastases from extramammary malignancies. Am Surg. 2004;70(4):287-290.
- Verger E, Conill C, Velasco M, Sole M. Metastasis in the male breast from a lung adenocarcinoma. Acta Oncol. 1992;31(4):479. doi:10.3109/02841869209088294
- Ucar N, Kurt OK, Alpar S, et al. Breast metastasis in a male patient with nonsmall cell lung carcinoma. South Med J. 2007;100(8):850-851. doi:10.1097/SMJ.0b013e3180f62fdc
- Fulciniti F, Losito S, Botti G, et al. Metastases to the breast: role of fine needle cytology samples. our experience with nine cases in 2 years. Ann Oncol. 2008;19(4):682-687. doi:10.1093/annonc/mdm546
- Maounis N, Chorti M, Legaki S, et al. Metastasis to the breast from an adenocarcinoma of the lung with extensive micropapillary component: a case report and review of the literature. Diagn Pathol. 2010;5:82. doi:10.1186/1746-1596-5-82
- Ji FF, Gao P, Wang JG, et al. Contralateral breast metastasis from pulmonary adenocarcinoma: two cases report and literature review. J Thorac Dis. 2012;4(4):384-389. doi:10.3978/j.issn.2072-1439.2012.02.03
- Mirrielees JA, Kapur JH, Szalkucki LM, et al. Metastasis of primary lung carcinoma to the breast: a systematic review of the literature. J Surg Res. 2014;188(2):419-431. doi:10.1016/j.jss.2014.01.024
- Fukumoto K, Usami N, Okasaka T, et al. Late breast metastasis from resected lung cancer diagnosed by epidermal growth factor receptor gene mutation. Lung Cancer. 2011;74(2):352-353. doi:10.1016/j.lungcan.2011.08.015
- Sato K, Takeyama Y, Yoshihara M, et al. CBDCA + pemetrexed + bevacizumab and its maintenance chemotherapy in a case of solitary breast metastasis from a lung adenocarcinoma resistant to gefitinib. Case Rep Oncol. 2012;5(3):546-553. doi:10.1159/000343678
- Dansin E, Carnot A, Servent V, et al. EGFR-mutated breast metastasis of lung adenocarcinoma: a case report. Case Rep Oncol. 2015;8(1):164-168. doi:10.1159/000381014
- Ota T, Hasegawa Y, Okimura A, et al. Breast metastasis from EGFR-mutated lung adenocarcinoma: a case report and review of the literature. Clin Case Rep. 2018;6(8):1510-1516. doi:10.1002/ccr3.1636
- Lin Q, Cai GP, Yang KY, et al. Case report: small cell transformation and metastasis to the breast in a patient with lung adenocarcinoma following maintenance treatment with epidermal growth factor receptor tyrosine kinase inhibitors. BMC Cancer. 2016;16;593. doi:10.1186/s12885-016-2623-4
- Martins RG, D’Amico TA, Loo BW Jr, et al. The management of patients with stage IIIA non-small cell lung cancer with N2 mediastinal node involvement. J Natl Compr Canc Netw. 2012;10(5):599-613. doi:10.6004/jnccn.2012.0062
- Albain KS, Swann RS, Rusch VW, et al. Radiotherapy plus chemotherapy with or without surgical resection for stage III non-small-cell lung cancer: a phase III randomised controlled trial. Lancet. 2009;374(9687):379-386. doi:10.1016/S0140-6736(09)60737-6
- van Meerbeeck JP, Kramer GWPM, Van Schil PEY, et al; European Organisation for Research and Treatment of Cancer—Lung Cancer Group. Randomized controlled trial of resection versus radiotherapy after induction chemotherapy in stage IIIA-N2 non-small-cell lung cancer. J Natl Cancer Inst. 2007;99(6):442-450. doi:10.1093/jnci/djk093
- Thomas M, Rübe C, Hoffknecht P, et al; German Lung Cancer Cooperative Group. Effect of preoperative chemoradiation in addition to preoperative chemotherapy: a randomised trial in stage III non-small-cell lung cancer. Lancet Oncol. 2008;9(7):636-648. doi:10.1016/S1470-2045(08)70156-6
- Higgins K, Chino JP, Marks LB, et al. Preoperative chemotherapy versus preoperative chemoradiotherapy for stage III (N2) non-small-cell lung cancer. Int J Radiat Oncol Biol Phys. 2009;75(5):1462-1467. doi:10.1016/j.ijrobp.2009.01.069
- de Cabanyes Candela S, Detterbeck FC. A systematic review of restaging after induction therapy for stage IIIa lung cancer: prediction of pathologic stage. J Thorac Oncol. 2010;5(3):389-398. doi:10.1097/JTO.0b013e3181ce3e5e
- Owen D, Chaft JE. Immunotherapy in surgically resectable non-small cell lung cancer. J Thorac Dis. 2018;10(suppl 3):S404-S411. doi:10.21037/jtd.2017.12.93
- Yi C, He Y, Xia H, et al. Review and perspective on adjuvant and neoadjuvant immunotherapies in NSCLC. Onco Targets Ther. 2019;12:7329-7336. doi:10.2147/OTT.S218321
- Raphael J, Vincent M, Boldt G, et al. Adjuvant epidermal growth factor receptor tyrosine kinase inhibitors (TKIs) in resected non-small cell lung cancer (NSCLC): a systematic review and meta-analysis. Am J Clin Oncol. 2019;42(5):440-445. doi:10.1097/COC.0000000000000533
- Huang Q, Li J, Sun Y, et al. Efficacy of EGFR tyrosine kinase inhibitors in the adjuvant treatment for operable non-small cell lung cancer by a meta-analysis. Chest. 2016;149(6):1384-1392. doi:10.1016/j.chest.2015.12.017
- Herbst RS, Tsuboi M, John T, et al. Osimertinib as adjuvant therapy in patients (pts) with stage IB-IIIA EGFR mutation positive (EGFRm) NSCLC after complete tumor resection: ADAURA. J Clin Oncol. 2020;38(18 suppl; abstr LBA 5). doi:10.1200/JCO.2020.38.18_suppl.LBA5
- Zhang H, Chen J, Liu T, et al. First-line treatments in EGFR-mutated advanced non-small cell lung cancer: a network meta-analysis. PLoS One. 2019;14(10):e0223530. doi:10.1371/journal.pone.0223530
- Nakagawa K, Garon EB, Seto T, et al; RELAY Study Investigators. Ramucirumab plus erlotinib in patients with untreated, EGFR-mutated, advanced non-small-cell lung cancer (RELAY): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019;20(12):1655-1669. doi:10.1016/S1470-2045(19)30634-5
Financial Disclosure: The authors have no significant financial interest in or other relationship with the manufacturer of any product or provider of any service mentioned in this article.

Oncology Peer Review On-The-Go: COVID-19, Cancer, and the Potential of mRNA Vaccines
March 30th 2021Mehmet Sitki Copur, MD, discussed his article in the Journal ONCOLOGY® focusing on COVID-19, messenger RNA vaccines, and the excitement surrounding its integration into the future of cancer treatment.
