A retrospective cohort study of women treated for early-stage breast cancer has confirmed rising trends in the proportion of patients who undergo mastectomy.

A retrospective cohort study of women treated for early-stage breast cancer has confirmed rising trends in the proportion of patients who undergo mastectomy.

Researchers have identified a mechanism that could explain how NSAIDs lower the risk of developing intestinal polyps that can transform into colon cancer.

A study found that prostate cancer patients with a history of heart problems are at increased risk of cardiac death following androgen-deprivation therapy.
Monotherapy with the investigational oral Bruton's tyrosine kinase inhibitor ibrutinib was well tolerated and produced durable responses in a phase Ib/II, open-label, multicenter study of 85 patients with relapsed/refractory chronic lymphocytic leukemia or small lymphocytic lymphoma.
Use of the bisphosphonate pamidronate (Aredia) may be “more efficient” than standard regimens as palliative treatment for symptoms of acute symptomatic osteonecrosis in pediatric patients with acute lymphoblastic leukemia.
Biennial visual inspection with acetic acid (vinegar) screening by trained public health workers significantly reduced cervical cancer mortality in a large cluster-randomized controlled trial conducted among thousands of women from poor neighborhoods in Mumbai, India.

Further analyses of data subsets from the ALSYMPCA study of the alpha particle-emitting isotope Ra-223 (Xofigo) were presented at ASCO, providing additional evidence of efficacy and safety of the recently FDA approved therapeutic agent.
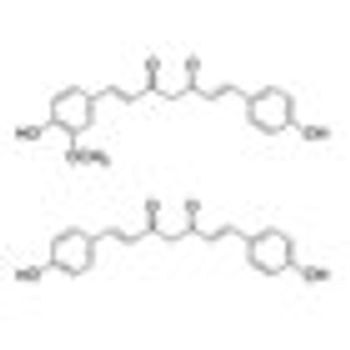
Addition of curcuminoids to treatment with docetaxel was well tolerated and showed promise in improving the response rate to docetaxel “in terms of both PSA decrease and objective response” in a phase II trial in patients with castration-resistant prostate cancer.
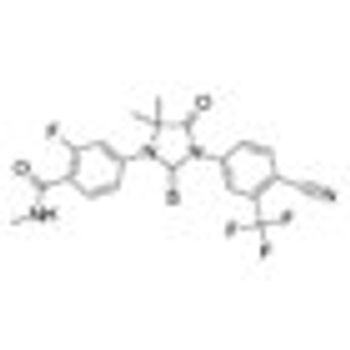
Monotherapy with enzalutamide (Xtandi) achieved a “high PSA response rate and marked PSA decline” in patients with hormone-naïve prostate cancer after 6 months in a single-arm, multicenter phase II study.
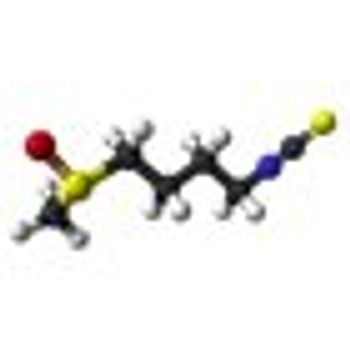
Treatment with 200 µmol per day of sulforaphane for 20 weeks was “feasible, safe,” and inhibited histone deacetylase (HDAC) function in a single-arm study of 20 patients who had non-castrate biochemical (PSA)-recurrence of prostate cancer despite surgery or radiation.
Findings suggesting that obesity substantially impairs the ability of L-asparaginase to kill leukemia cells were reported last month by investigators from The Saban Research Institute, Children’s Hospital Los Angeles.
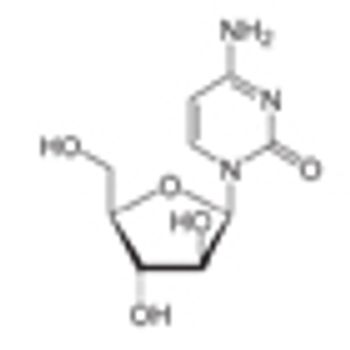
Targeting the bone marrow–specific extracellular matrix osteopontin may be an effective strategy to increase the efficacy of chemotherapy in patients with acute lymphocytic leukemia, according to the authors of a recent study.
A dose-adjusted regimen of etoposide, doxorubicin, and cyclophosphamide with vincristine, prednisone, and rituximab (DA-EPOCH-R) obviated the need for radiotherapy in patients with primary mediastinal B-cell lymphoma in a single-group, phase II, prospective study.

An ongoing phase III study comparing the efficacy and safety of perifosine in patients with relapsed or relapsed/refractory multiple myeloma has been discontinued.

Researchers have discovered a fully human “T-cell receptor–like” monoclonal antibody specific for the Wilms tumor 1 oncoprotein-an intracellular oncogenic transcription factor that is overexpressed in a wide range of leukemias, as well as solid cancers.
Combination chemotherapy with sequential administration of methotrexate and asparaginase was found to be effective salvage therapy in a small retrospective study of pediatric patients with relapsed or refractory acute myeloid leukemia (AML).

Ado-trastuzumab emtansine, formerly known as T-DM1, improved progression-free survival as first-line treatment for patients with HER2-positive metastatic breast cancer, when compared with the standard treatment of trastuzumab plus docetaxel in a randomized phase II multicenter study.
Findings of a newly published study by an international team of researchers suggest that targeting growth factor independence 1 (GFI1) may improve the prognosis of patients with T-cell acute lymphoblastic leukemia or other lymphoid leukemias.
Researchers from the University of Rochester have identified BCL2 inhibitors as potential leukemia stem cell-targeting agents, demonstrating that two such inhibitors killed inactive and metabolically slower leukemia stem cells.
A single tube 10-color assay for the detection of residual disease in chronic lymphocytic leukemia provides at least equivalent results to the international standardized flow cytometry approach to detection and requires fewer cells, uses fewer reagents, and allows for simpler analysis, according to a new study.
The combination of two antibody-based targeting agents-inotuzumab ozogamicin and rituximab-demonstrated high response rates and long progression-free survival in patients with relapsed follicular lymphoma or relapsed diffuse large-B-cell lymphoma (DLBCL) in an international phase I/II multicenter, open-label study.
A combination of all-trans retinoic acid (ATRA) and arsenic trioxide resulted in outcomes “at least not inferior” to those achieved when ATRA was used in combination with idarubicin chemotherapy in newly diagnosed patients with non–high-risk, acute promyelocytic leukemia.
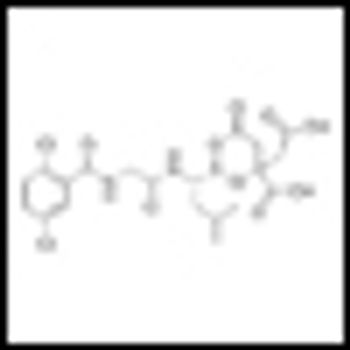
Preliminary findings presented at ASH suggest a “favorable emerging clinical profile” for once weekly administration of MLN9708 in combination with lenalidomide and dexamethasone in patients with newly diagnosed multiple myeloma.
ARRY-520, a novel selective kinesin spindle protein (KSP) inhibitor showed promising clinical activity both alone and combined with low-dose dexamethasone in a phase II clinical trial in patients with relapsed and refractory multiple myeloma.

Updated findings from the pivotal phase II PACE trial show sustained benefit of the investigational BCR-ABL tyrosine kinase inhibitor ponatinib in heavily pretreated patients with resistant or intolerant chronic myeloid leukemia or Philadelphia chromosome-positive acute lymphoblastic leukemia.
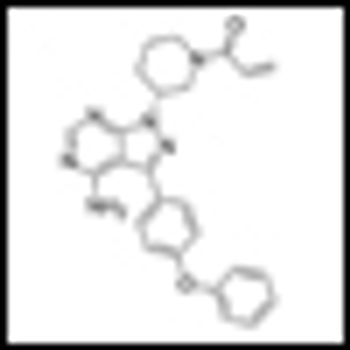
Impressive interim results from a phase Ib/II trial of ibrutinib, a selective Bruton tyrosine kinase inhibitor, in patients with chronic lymphocytic leukemia (CLL) were reported at ASCO.
Updated clinical data from the pivotal phase II global PACE trial of ponatinib confirm its impressive antileukemic activity in patients with chronic myeloid leukemia or Philadelphia-chromosome-positive acute lymphoblastic leukemia at all stages who are resistant or intolerant to dasatinib or nilotinib.
Bendamustine plus rituximab should be considered as the preferred first-line treatment of follicular and indolent lymphomas, and the elderly with mantle cell lymphoma.
Updated data from an ongoing phase II trial of oral sapacitabine showed that the drug had activity in older patients with myelodysplastic syndromes refractory to front-line hypomethylating agents indicates.
In a phase I study the targeted drug crizotinib, a small molecule inhibitor of anaplastic lymphoma kinase and met proto oncogene, delayed or eliminated signs of tumor growth in pediatric patients with aggressive cancers.
December 12th 2011