
Patients with BRCA-mutated breast cancer can safely undergo assisted reproductive techniques, according to an ESMO study.

Patients with BRCA-mutated breast cancer can safely undergo assisted reproductive techniques, according to an ESMO study.
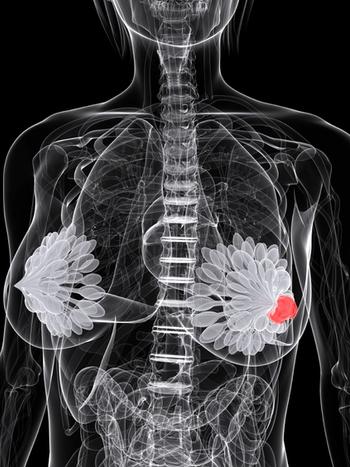
Updated findings from the phase 3 MONALESSA-3 trial identified a significant overall survival benefit among postmenopausal patients with hormone receptor–positive, HER2-negative breast cancer who were treated with ribociclib and fulvestrant vs matched placebo.
CancerNetwork® spoke with Dr. Sara Hurvitz, associate professor of medicine at the University of California, Los Angeles, about therapy options for breast cancer.

CancerNetwork® spoke with Dr. Peter Schmid about the KEYNOTE-522 trial.
Dr. Rebecca Dent, oncologist at the National Cancer Center, Singapore, discusses the rationale for immunotherapy in early-stage breast cancer.
Dr. Leisha Emens of the University of Pittsburgh Medical Center's Hillman Cancer Center discusses the overall survival in the KATE2 study.
Two studies from ESMO have potentially found treatments which can improve survival in HR-positive breast cancer.
Adding the immunotherapy pembrolizumab to neoadjuvant chemotherapy improved the pathological complete response rate in patients with early triple-negative breast cancer, according to findings of a study presented at ESMO.