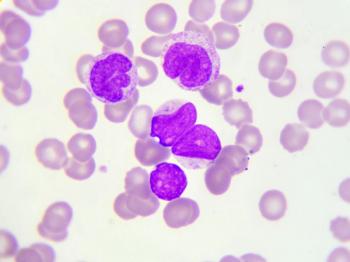
A retrospective study evaluating over 3.9 million children found that Down syndrome was a strong risk factor for the development of childhood leukemia and has a stronger association with acute myeloid leukemia than previously recorded.

Your AI-Trained Oncology Knowledge Connection!


A retrospective study evaluating over 3.9 million children found that Down syndrome was a strong risk factor for the development of childhood leukemia and has a stronger association with acute myeloid leukemia than previously recorded.
The NanoTherm therapy system for focal ablation of intermediate-risk prostate cancer continued to show a tolerable safety profile in a recent analysis.

Adding daratumumab led to at least a very good partial responses in all 41 patients receiving weekly carfilzomib, lenalidomide, and dexamethasone, and complete responses in all but 2 patients.

CancerNetwork® shares its latest investigation into novel practices for rendering surgical management of prostate cancer from experts at Vanderbilt Institute for Surgery and Engineering.

Look back at some of the important news and notes from last week in the world of oncology, featuring articles from live events, the latest issue of the Journal ONCOLOGY®, and an episode of “Oncology Peer Review On-The-Go.”
A phase 3 trial found that pembrolizumab for patients with microsatellite instability–high or mismatch repair–deficient metastatic colorectal cancer significantly improved health-related quality of life compared with chemotherapy.
Rolling submission with the FDA for surufatinib treatment in patients with pancreatic and extra-pancreatic neoendocrume tumors was completed and an expanded access program for the drug is currently underway for patients in the United States.
Recent guidelines have advised against routine use of sentinel lymph node biopsy and radiotherapy in patients over age 70 years with breast cancer, but a new study finds most patients still receive the interventions.
CancerNetwork® spoke with Neelam and Sanju Sinha of the National Cancer Institute about their research into sex differences associated with using tumor mutational burden to predict response to PD-1 inhibition.
Data in the Journal of Clinical Oncology found that low-intensity chemotherapy without additional intensified pegaspargase cured almost all patients treated on the Children’s Oncology Group AALL0331 trial.

The phase 2 trial showed that tipifarnib was effective for patients with metastatic head and neck squamous cell carcinoma who have limited therapeutic options available.
CancerNetwork® spoke with Francesco Ravera, MD, PhD, during the American Association for Cancer Research Annual Meeting 2021 to discuss results of a study aimed at determining pathological complete response in patients with locally advanced breast cancer by either cell-free DNA assessment or traditional MRI.
The interim analysis showed positive response and safety data in adult patients with relapsed or refractory CLL or SLL treated with zanubrutinib compared with those receiving ibrutinib.

The lead author spoke about the rising incidence of early-onset colorectal cancer, which was the focal point of her research presented at the American Association for Cancer Research Annual Meeting 2021.
The phase 2 trial investigated the toxic effects, PSA responses, and resulting quality of life measures from SBRT and SDRT, finding that the latter has the potential to be a feasible alternative treatment option for prostate cancer.

Based on results of the recently reported phase 3 CheckMate 274 trial, the FDA has granted priority review designation to nivolumab for the adjuvant treatment of high-risk muscle-invasive bladder cancer.

By a close decision, the FDA’s Oncologic Drugs Advisory Committee voted against upholding the accelerated approval of nivolumab monotherapy to treat patients with hepatocellular carcinoma following sorafenib.

Marron spoke about the value that neoantigen vaccines can provide by studying T-cell responses and the characteristics of lymphoid response to antigens.
CancerNetwork® spoke with Francesco Ravera, MD, PhD, during the American Association for Cancer Research Annual Meeting 2021 to discuss a study that looked at cell-free DNA assessment compared with traditional MRI for determining pathological complete response in patients with locally advanced breast cancer.
Data examining thermal ablation for women with a total of 119 metastatic gynecologic tumors found the therapy was safe and effective in the local control of these tumors.

Chemotherapy-induced myelosuppression was reduced when trilaciclib was used before chemotherapy for patients with extensive-stage small cell lung cancer versus placebo.

Accelerated approval for atezolizumab in the treatment of patients with locally advanced or metastatic urothelial carcinoma who are not eligible for certain chemotherapy regimens was maintained by an FDA committee vote.

Better adherence to oral chemotherapy was seen in patients who used a smartphone mobile app, according to a presentation at the Oncology Nursing Society’s 46th Annual Congress.

CancerNetwork® sat down with Health and Wellness expert Sonia Jhas to discuss best practices for avoiding unhealthy eating habits on a demanding schedule.

Based on response efficacy in a phase 1/2 trial, the FDA granted the selective oral tyrosine kinase inhibitor mobocertinib priority review for patients with pretreated metastatic non–small cell lung cancer harboring insertion mutations in EGFR exon 20.

The FDA’s Oncologic Drugs Advisory Committee voted 7 to 2 in favor of atezolizumab plus nab-paclitaxel maintaining its accelerated approval to treat patients with unresectable, locally advanced or metastatic triple-negative breast cancer (TNBC) whose tumors express PD-L1.

Kim discussed the need for further CREB inhibitor combinations to target collateral pathways and improve patient outcomes.

CancerNetwork’s latest podcast episode dissects and article from the journal ONCOLOGY® focusing on 4 PARP inhibitors investigated for the treatment of advanced prostate cancer.
A phase 2 trial found that acalabrutinib may present a potential therapeutic option for patients with chronic lymphocytic leukemia who discontinued ibrutinib treatment.
Low-grade, manageable treatment-emergent adverse effects and low rates of discontinuation were associated with the RET inhibitor selpercatinib as therapy for patients with RET-altered advanced solid tumors who were treated on the LIBRETTO-001 trial.