A new analysis of the NLST showed that both the benefits and harms of low-dose CT screening are slightly greater among patients older than 65 years.

A new analysis of the NLST showed that both the benefits and harms of low-dose CT screening are slightly greater among patients older than 65 years.

Patients overdue for a screening mammography were more likely to return for an appointment when they received a reminder postcard and a physician signed letter.

Using a simple tool to measure performance status can help physicians predict survival and facilitate end-of-life planning in outpatients with advanced cancer.

When someone states they are “tired,” it prompts follow-up questions regarding activity, sleep, diet, and stress, among other things-just as a complaint of pain leads to where, what kind, and how long.
Survivors of Hodgkin lymphoma may be at increased risk of diabetes if they were exposed to radiation to the para-aortic lymph nodes and spleen during treatment.

When communicating with the difficult patient doctors are advised to avoid such approaches as snapping, bristling, or even a subtle curling of the lips.
Patients with limited life expectancies often undergo routine cancer screenings despite evidence that they are unlikely to benefit and may experience harm.

The treatment of inoperable stage III non–small-cell lung cancer (NSCLC) remains a challenge due to high rates of distant metastasis, local recurrence, and toxicity associated with definitive therapy.

A large European study found that PSA screening reduces prostate cancer deaths, but that these benefits do not outweigh the harms of overtreatment.

Recently, my treatment center changed the contrast agent patients are asked to drink prior to having a CT. They look and taste the same. So why has the switch bothered me so much?

Patients with Medicaid coverage or no insurance at all were more likely to present with advanced cancer of a number of types, and were also less likely to receive certain treatments, according to a study using the SEER database.

Each person facing cancer has their own way of coping. They have no obligation to fit a stereotype that others may have conjured up. They are each the poster child of their own unique campaign.

A 3-year pilot program that rewarded oncologists for improving outcomes and efficiency-rather than administering drugs-significantly reduced overall costs without lowering quality of care.

End-of-life care is challenging for even the most seasoned oncologist. Here are five suggestions that can help you better navigate this difficult but critical part of your work.
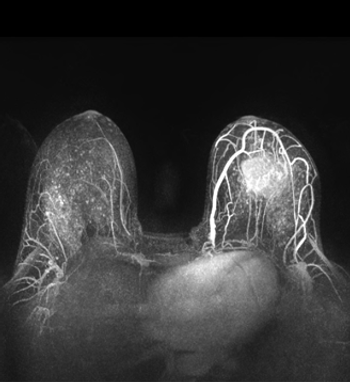
An annual screening for breast cancer using a combination of MRI and mammography could be an effective program for high-risk women, particularly those who are known BRCA mutation carriers.

In light of the recent FDA approval of HPV testing for women as a screening method for cervical cancer, we discuss changing guidelines with two experts.

Regardless of where you practice, good communication between you and your pathologist is the best way to ensure that correct testing is done. Here are six common points of miscommunication to watch out for.
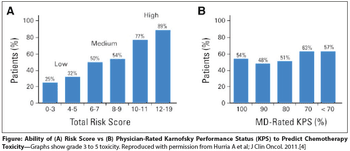
Making appropriate treatment decisions for older adults with cancer is one of the most important challenges that oncologists face in daily practice, as the therapy selected depends on an assessment of the patient’s “fitness.”

When the media reported earlier this year that giant drugstore chain CVS had announced that it would stop selling tobacco products, it appeared to be a sudden, independent, and ethically responsible business decision. In fact, there is important background and subtext.
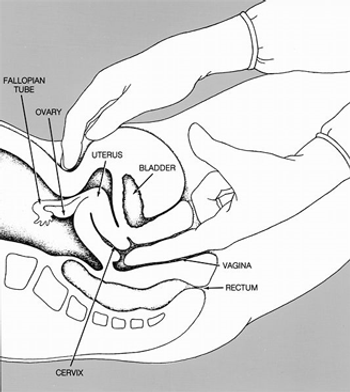
A new clinical practice guideline from the American College of Physicians recommends against conducting routine pelvic exams in average-risk, asymptomatic women.

Researchers have developed a tool to measure patients’ risk for financial toxicity following cancer diagnosis and treatment.

In this interview, Dr. Hyman Muss discusses some of the barriers to providing high-quality care to geriatric cancer patients, including the barriers to diagnosis and treatment.
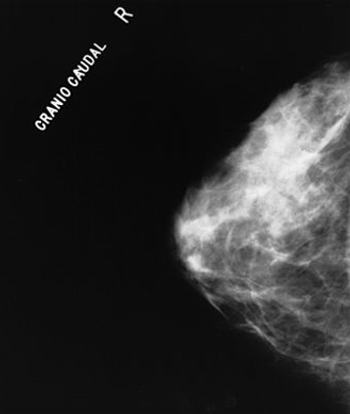
The combination of four imaging techniques together results in the ability to better differentiate between benign and malignant breast tumors, which may lead to fewer unnecessary breast biopsies.

Is there any truth in advertising? A recent study found that cancer center ads only emphasize positive outcomes, but other than direct-to-consumer marketing of pharmaceuticals that list all possible side effects, are you aware of any ads that state less than optimal outcomes?

Oncologists who participated in communication workshops improved their ability to talk with patients about their prognoses and provide emotional support, a recent study found.

The CALGB 369901 trial examined how frailty and older age influence the use of adjuvant hormonal therapy for breast cancer and found that while frailty can deter the start of therapy, frail patients who had started on a regimen were not much more likely to discontinue their treatment early.
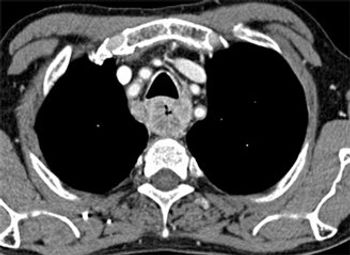
Preoperative staging of esophageal cancer with PET/MR is approximately as good as with endoscopic ultrasound, and may improve slightly over ultrasound and PET/CT scans, according to a new study.

There is a need to learn more about the effect of hypofractionation on an individual patient’s breast cancer outcomes and tissue toxicities, based on both biologic and technical variables, so that the treatment decision is not primarily a matter of dollars and cents.

Customized more aggressive treatments should be given to patients with the worst prognosis. For most of the other breast patients, shorter and often milder treatment is also a humble victory in our daily struggle against cancer.

Barriers to cost discussions fall into three categories: inaccessible cost data, ethical concerns, and insufficient training.