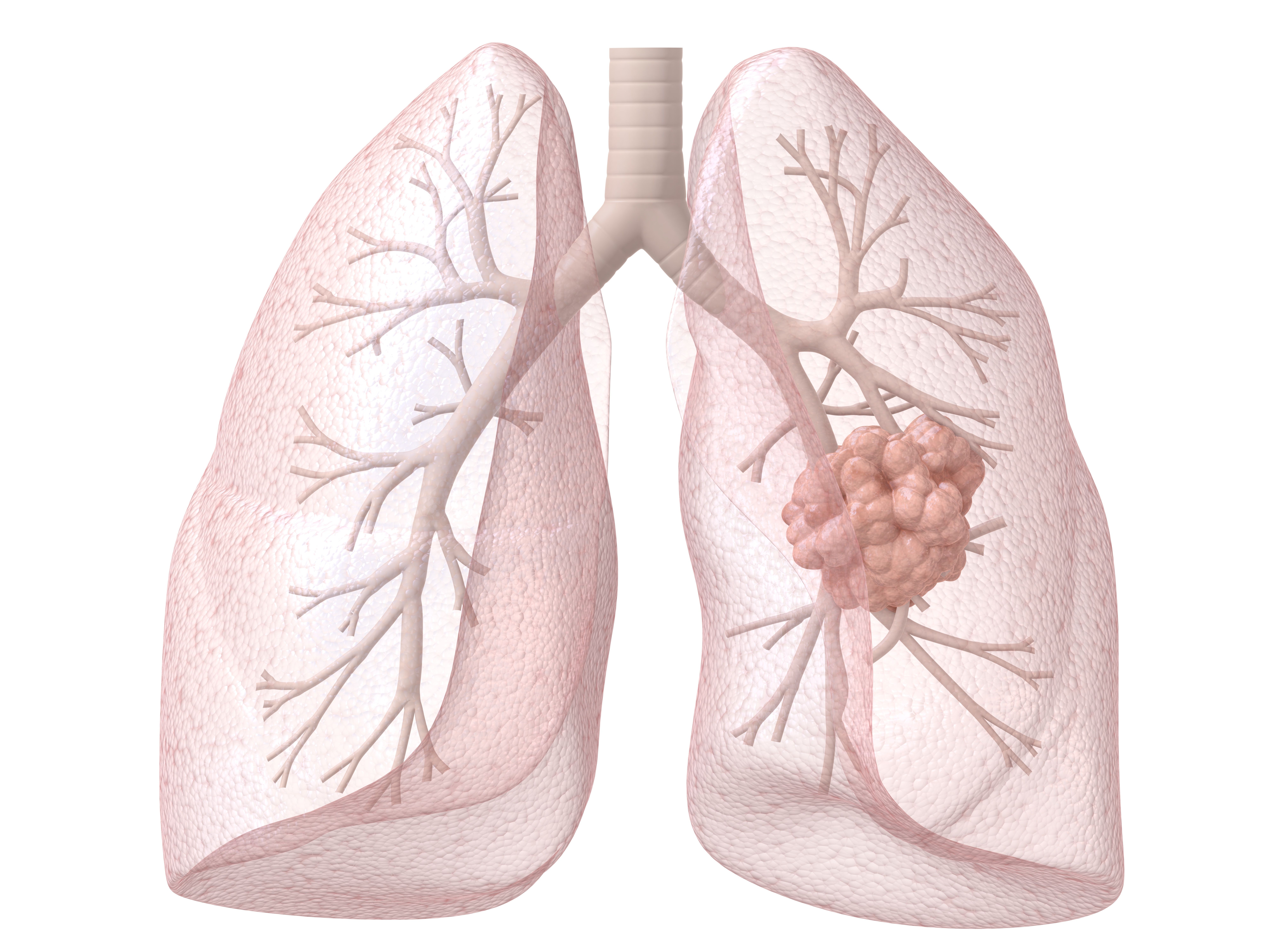
Lung Cancer
Latest News
Video Series

Latest Videos
Shorts








Podcasts
More News

The FDA approved zongertinib in HER2-mutated NSCLC as part of its Commissioner's National Priority Voucher pilot program.
Read this discussion to spot LEMS red flags tied to small cell lung cancer, avoid misdiagnosis, and follow antibody/EMG testing and screening steps.

Olvimulogene nanivacirepvec yielded 3 partial responses among 9 evaluable patients with SCLC who had progression or recurrence on prior therapy.

OncoPrism-NSCLC was found to be a predictive biomarker of immune checkpoint inhibitor benefit vs a general prognostic tool in patients with NSCLC.
Expert teams debate cutting-edge lung cancer treatments, exploring trial data and innovative strategies for improving patient outcomes in NSCLC and SCLC.

An oncologist at the Georgia Cancer Center discussed the evolution of treatment strategies and emerging therapies for patients with EGFR-mutated disease.
The blood-based test detected 31% of lung cancers one year prior to in-trial diagnosis compared with 8% of cancers identified by low-dose CT or Lung-RADS.

Advancements in antibody drug conjugates, bispecific therapies, and other targeted agents may hold promise in lung cancer management.

Stressing the importance of prompt AE disclosure before they become severe can ensure that a patient can still undergo resection with curative intent.

Thomas Marron, MD, PhD, presented a session on clinical data that established standards of care for stage II and III lung cancer treatment at CFS 2025.

Data from the DeLLphi-304 trial support the full approval of tarlatamab in this extensive-stage small cell lung cancer population.
One patient with metastatic bladder cancer experienced an ongoing metabolic complete response following treatment with aldesleukin/imneskibart.
Data from a phase 1a/1b trial show that no patients discontinued STK-012 due to treatment-related adverse effects.

Dato-DXd is being assessed in numerous trials across the breast, lung, and bladder cancer spaces.

Decreasing the low-dose bath of proton therapy to the body may limit the impact of radiation on lymphocytes and affect tumor response.

ILKN421H plus pembrolizumab previously showed antitumor activity among patients with frontline non–small cell lung cancer in a phase 1 trial.

According to Eyub Akdemir, MD, reducing EDIC may be feasible without compromising target coverage to reduce anticipated lymphopenia rates.
Data from the phase 2 TUXEDO-3 trial support patritumab deruxtecan as a novel treatment option across different cancer populations with brain metastases.

Data from the REZILIENT2 trial show meaningful intracranial activity in patients with NSCLC harboring EGFR exon 20 insertions or other uncommon mutations.

Although survival outcomes were numerically improved with ROS1-targeted therapies, an unmet need remains for patients with ROS1-mutated advanced NSCLC.

Alectinib exhibited a CNS DFS improvement, with a 63% reduction in the risk of this event, and 4-year CNS DFS rate was 90.4% vs 76.1% with chemotherapy.
Retrospective cohort findings may inform tailored treatment approaches for frontline metastatic BRAF V600E–mutated non–small cell lung cancer.
Data from the KEYNOTE-671 trial support the use of pembrolizumab among patients with non–small cell lung cancer in the perioperative setting.

Data from the phase 3 HARMONi-6 study may support ivonescimab plus chemotherapy as a new standard of care in advanced squamous non–small cell lung cancer.
Slow accrual led to the early stopping of the phase 3 DREAM3R trial assessing durvalumab/chemotherapy in advanced pleural mesothelioma.