Poll|Poll|May 11, 2012
A 54-Year-Old Woman Notes the Abundant Development of Nonpigmented Hair on Her Ears and Nose
Author(s)Ted Rosen, MD
Advertisement
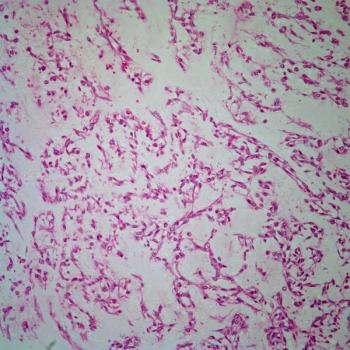
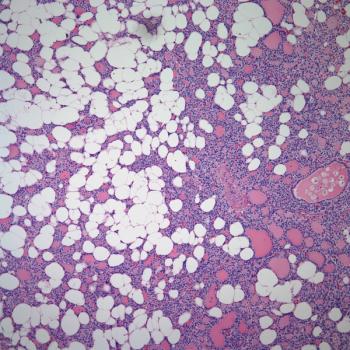
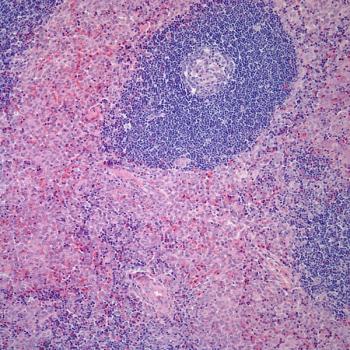
A 54-year-old woman, previously in excellent health, noted the insidious development of abundant, thin, and softly textured, nonpigmented hair on her ears and nose. A detailed review of systems was entirely negative, and both a complete blood count and automated routine biochemistry panel were normal. Gynecologic examination, including a pap smear, was normal one year prior to presentation. The patient was a lifelong nonsmoker and nondrinker. Family history was negative for cancer.
From what specific disorder does she suffer?
Newsletter
Stay up to date on recent advances in the multidisciplinary approach to cancer.
Advertisement
Latest CME
Advertisement
Advertisement
Trending on CancerNetwork
1
Anbenitamab Injection Combo Meets pCR End Point in HER2+ Breast Cancer
2
CAR-T Moves to the Front Line: Rethinking the Multiple Myeloma Sequence
3
Urine-Based Biopsy Shows 97.8% Sensitivity in Prostate Cancer Detection
4
ELCC 2026: Key Updates Across Non–Small Cell Lung Cancer Populations
5