
Findings from the phase 3 EV-304 trial support the supplemental biologics license application for enfortumab vedotin/pembrolizumab in patients with MIBC.

Findings from the phase 3 EV-304 trial support the supplemental biologics license application for enfortumab vedotin/pembrolizumab in patients with MIBC.

Presenters at the 3rd Biennial Miami Precision Medicine 2026 Conference shared insights related to pancreatic cancer, sarcomas, and genitourinary diseases.

Data from the Beamion LUNG-1 trial may help clinicians make informed decisions on HER2-targeted therapy for those with advanced or metastatic NSCLC.

Iberdomide and mezigdomide have achieved positive phase 3 results in multiple myeloma, according to Shebli Atrash, MD.

Elizabeth Mittendorf, MD, discussed how the Clarity Breast AI algorithm can complement tools such as the Tyrer-Cuzick model to define breast cancer risks.

Orca-T has demonstrated clear benefits in reducing graft-vs-host disease among patients with hematologic malignancies, according to Wendy Stock, MD.

All injections of JNJ-1900 have been completed according to plan so far in the phase 2 CONVERGE study, said Benjamin Cooper, MD.

Constant adjustments during high-intensity focused ultrasound may help minimize damage to healthy tissue in prostate cancer treatment.

Experts discuss the “transformational” potential of enfortumab vedotin plus pembrolizumab for patients with muscle-invasive bladder cancer.

Sarah Weiss, MD, discussed key IGNYTE trial findings of RP1 and nivolumab in patients with advanced melanoma who progressed on prior anti-PD-1 therapy.

Heath Skinner, MD, PhD, discussed current standards for radiotherapy in head and neck cancers and highlighted multidisciplinary collaboration within UPMC.

Zenocutuzumab has been added to the NCCN’s Clinical Practice Guidelines in Oncology for biliary tract cancers based on results from the phase 2 eNRGy trial.

Among patients with recurrent cervical cancer who received prior immunotherapy, sac-TMT plus pembrolizumab achieved an ORR of 54%.

Combining tisotumab vedotin with carboplatin, pembrolizumab, and optional bevacizumab produces robust responses in first-line recurrent or metastatic cervical cancer.

Roisin E. O’Cearbhaill, MD, discussed the MUC16 × CD28 bispecific antibody REGN5668 in treating patients with recurrent ovarian or endometrial cancer.

Although ctDNA has limited utility in well-differentiated disease, it may have use in monitoring molecular residual disease in high-grade carcinomas.

The primary and secondary end points of the TANGENT trial were met when patients with tenosynovial giant cell tumor were treated with emactuzumab.
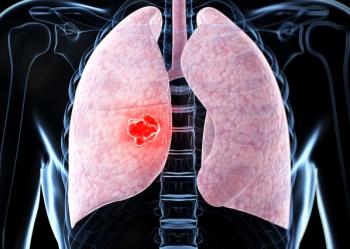
The FDA has assigned a Prescription Drug User Fee Act date of October 10, 2026, for ifinatamab deruxtecan in extensive-stage small cell lung cancer.
Although not yet mature, overall survival data trended in favor of pirtobrutinib plus venetoclax/rituximab in the phase 3 BRUIN CLL-322 trial.

The FDA has granted FTD and cleared an IND application for daretabart, an anti-GD2 monoclonal antibody, for the treatment of high-risk neuroblastoma.
Involving transplant physicians early on is critical in myelofibrosis therapy, said Nicolaus Kröger, MD.

Phase 1 data show that anti-BCMA CAR-T cells can be generated directly within a patient without a need for leukapheresis and lymphodepleting chemotherapy.

Results from the phase 3 RASolute 302 trial showed that daraxonrasib improved median OS compared with standard chemotherapy in metastatic PDAC.

Elizabeth Mittendorf, MD, PhD, discusses Clarity Breast, an FDA-authorized AI tool that predicts 5-year breast cancer risk using standard 2D mammograms.

At the MRD assessment on day 45, patients with large B-cell lymphoma treated with cema-cel saw a median decrease of 97.7% in their plasma ctDNA levels.

Among all patients with gynecologic malignancies who received a formal radiation cystitis diagnosis, 87.5% received treatment and 56.0% were referred to a urologic specialist.
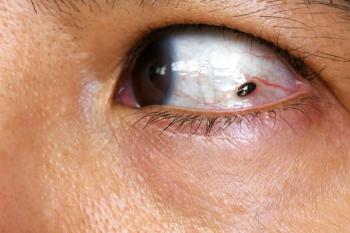
PFS showed statistically significant improvement with darovasertib plus crizotinib in the first line for patients with HLA-A*A2:01–negative metastatic uveal melanoma.

Young patients with breast cancer should always aim to advocate for themselves, said Shari Goldfarb, MD.

Across both pretreated cervical cancer and PROC populations, no new safety signals and low rates of treatment discontinuation were observed with DB-1311/BNT324.

Making pancreatic cancer an immunotherapy-receptive disease requires identifying select patient subgroups who may benefit.