
Data from the PRESERVE-003 trial may support the use of a novel chemotherapy-free option for patients with squamous non–small cell lung cancer.

Data from the PRESERVE-003 trial may support the use of a novel chemotherapy-free option for patients with squamous non–small cell lung cancer.

The FDA has granted orphan drug designation to tovecimig (CTX-009) for the treatment of bile duct cancer, following results from the phase 2/3 COMPANION-002 trial.

The RUBY trial demonstrates that disrupting the BCL11A binding site in the γ-globin promoters can normalize hemoglobin in severe sickle cell disease.

According to Jeff P. Sharman, MD, determining whether a patient requires inpatient or outpatient admission is a critical concern in community practice.

Data from the phase 1/2 ALKOVE-1 trial support the application for neladalkib in this non–small cell lung cancer population.

Natalie Berger, MD, discusses the evolving landscape of ADC sequencing in breast cancer, the role of HER2 expression, and the "sandwich approach" to treatment.

Most treatment-related adverse effects were grades 1 or 2, and no grade 4 adverse effects or treatment-related deaths were observed with Lu-PSMA-617.

Data from a phase 3 trial may support the role of carfilzomib in the upfront treatment setting for patients with multiple myeloma.

The 2026 ASCO thyroid cancer guideline update focused on the shifting landscape of systemic therapy for advanced disease.

Experts discussed the critical intersection of oncology and mental health, which included identifying and mitigating suicide risk factors.

Teclistamab/daratumumab is one of the most exciting combinations seen in relapsed/refractory multiple myeloma, said María-Victoria Mateos, MD, PhD.

Mina Fam, MD, explored the integration of multiparametric MRI and genomics in focal HIFU selection and compared its AE profile to radical intervention.

Frequent hydration, salivary gland stimulation, and overnight humidifier use are strategies Bradley J. Stish, MD, employs to manage dry mouth.

Data from the PALOMA-2 trial showed comparable efficacy with subcutaneous amivantamab vs prior reports of the intravenous formulation.

Robert Coleman, MD, FACOG, FACS, discusses the significance of the relacorilant and nab-paclitaxel approval in PROC supported by phase 3 ROSELLA trial data.

Although numerous bispecific agents are FDA approved for large B-cell lymphoma, many did so based on single-arm studies requiring confirmation studies.

Data demonstrate that reprogramming immunosuppressive macrophages can convert cold tumors into self-sustaining immune battlefields without lymphodepletion.
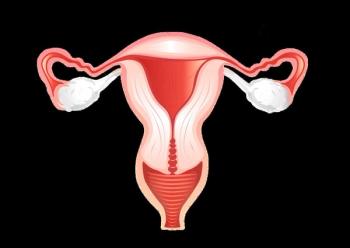
The FDA granted FTD to CTIM-76, a CLDN6/CD3 bispecific antibody, for the treatment of patients with CLDN6-positive platinum-resistant ovarian cancer.
Poster presentations at ELCC 2026 highlighted potential advances in neoadjuvant immunotherapy, intratumoral radioenhancers, and other modalities.

Continuing to implement prophylactic measures is necessary to maintain the benefit of subcutaneous amivantamab plus lazertinib in this NSCLC population.

The CAREMM-001 study may offer a glimpse into a potential future in which the therapeutic sequence of multiple myeloma is fundamentally reimagined.

Bradley J. Stish, MD, emphasized the role of decision-making processes between physicians and patients when deciding between 2.5 and 5 mg of oxybutynin.

Results from a phase 1/2a trial and the ongoing FORTE basket study support the FDA’s regulatory decision for these patients with recurrent or progressive glioma.

The phase 3 EMERALD-3 trial met its primary end point of PFS with durvalumab, tremelimumab, lenvatinib, and TACE in patients with unresectable HCC.

Investigators of a prospective validation study highlighted the potential of a urine-based assay to reduce surgical biopsies deemed unnecessary.

Management strategies for oxybutynin-induced xerostomia include hydration, sugar-free gum, and humidification.

The totality of evidence from the phase 2 ZUMA-2 study formed the basis of the regulatory decision.

The FDA has extended the PDUFA action date to July 6, 2026, for approving Orca-T across different hematologic malignancies.


Ryan H. Moy, MD, PhD, is the principal investigator of a phase 2 trial evaluating defactinib plus avutometinib in patients with diffuse gastric/GEJ carcinoma.