
Given that prostate cancer is the most common non-cutaneous cancer among men in the Western world, its treatment is of great medical and public significance.

Given that prostate cancer is the most common non-cutaneous cancer among men in the Western world, its treatment is of great medical and public significance.

In the current critical review we discuss these emerging trends in localized and systemic treatment as well as possible interesting combinations of the two modalities. Finally, we discuss the role of the new systemic agents in non–clear cell RCC.

Researchers reported results of an international phase III trial showing that intermittent hormonal therapy is less effective than continuous therapy in prostate cancer with minimal disease.
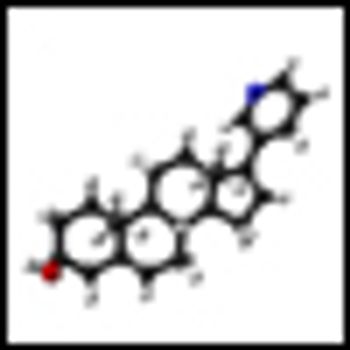
Results from a randomized phase II study show that 6 months of treatment with abiraterone (Zytiga), in addition to the hormone therapy leuprolide before prostatectomy, resulted in complete response and near-complete response in one-third of men with high-risk prostate cancer.
In a multicenter phase III trial of 360 patients with muscle-invasive bladder cancer, synchronous chemoradiotherapy provided better locoregional control without significant added toxicity, investigators for the Bladder Cancer 2001 trial have found.

Two experts discuss newly available and upcoming treatment options, such as abiraterone and MDV3100, for patients with castration-resistant prostate cancer.

Radium-223 is a promising agent that represents a new class of alpha pharmaceuticals that gets down to the site of bony metastases. The limited side-effect profile potentially allows for repeat administration to increase durability of pain control, and for its use in combination with novel biologic and chemotherapeutic agents.

One would hope that survival data from at least one more phase III or phase IV clinical trial will convincingly show a prolongation of survival due to treatment with Alpharadin. This will not be inexpensive therapy.

This article will present current information about alpha-pharmaceuticals, a new class of targeted cancer therapy for the treatment of patients with CRPC and bone metastases. It will review preclinical and clinical studies of the experimental radiopharmaceutical radium-223 chloride (Alpharadin).

Use of neoadjuvant metformin prior to radical prostatectomy helped to reduce metabolic effects and slow the growth rate of cancer in a single-center phase II study conducted among men with confirmed prostate cancer.
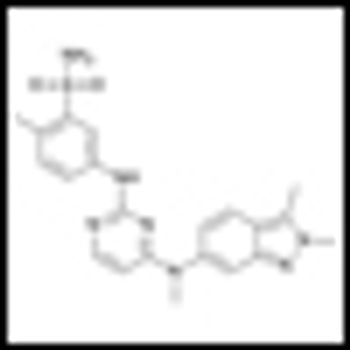
The antiangiogenic agent pazopanib demonstrated clinically meaningful activity in patients with refractory urothelial cancer in a phase II proof-of-concept study, identifying pazopanib as the first targeted compound to have clinically meaningful activity in patients with refractory urothelial cancer.

Rationale for prostate cancer screening continues to be debated as an update to a large European trial reconfirms a reduction in death rates from prostate cancer in men who are screened for the disease. The study, however, found no significant difference in overall mortality between the two arms of the trial.

Renal cell carcinoma (RCC) had historically been regarded as a disease that was refractory to therapy once surgical options had been exhausted.

The development of vascular endothelial growth factor (VEGF) pathway inhibitors and mammalian target of rapamycin (mTOR) inhibitors for the treatment of renal cancer is a real success story.

The article by Posadas and Figlin on systemic therapy in advanced renal cell carcinoma (RCC) provides a very interesting and comprehensive review of our current knowledge concerning the treatment of RCC.
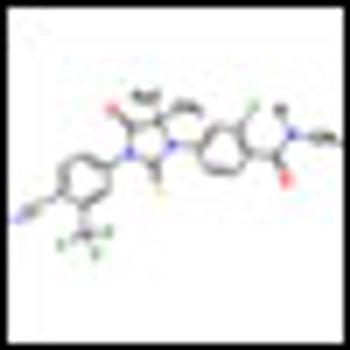
The new drug, MDV3100, extended overall survival by 4.8 months (P < .001) and reduced the risk for death by 37% as compared to placebo in men with castration-resistant prostate cancer who had progressed after treatment with docetaxel.

Initial results of a multicenter trial show that 2 biomarkers, PCA3 and T2-ERG, are found at high levels in prostate cancer compared to noncancerous prostate cells and correlate well with 2 indicators of aggressive prostate cancer, tumor volume and Gleason score.

CancerNetwork speaks with Dr. Michael Atkins, who has extensive clinical experience in kidney cancer and development of various new treatments and is presenting this weekend during the renal cancer translational science session at the American Society of Clinical Oncology 2012 Genitourinary Cancers Symposium.
It is still difficult to gauge the probability that a low-risk prostate cancer patient may be upgraded to a higher prostate cancer stage. Researchers at Vanderbilt University Medical Center have now determined that smaller prostates were more likely to evolve into more serious, aggressive disease.

The US Food and Drug Administration (FDA) has approved the angiogenesis blocker axitinib (Inlyta), a twice daily oral drug, as a second-line treatment for patients with advanced renal cell carcinoma.

After several decades with only modest changes in the therapeutic paradigm, rapid progress in understanding the biology of advanced prostate cancer has been translated into more accurate terminology, such as “castration-resistant” (as opposed to “hormone-refractory” or “androgen-independent”) prostate cancer, as well as clinically meaningful therapeutic developments.

These are, indeed, exciting times for patients with metastatic prostate cancer and the clinicians who care for them.

We have entered a period of accelerated drug development and optimism in the care of advanced prostate cancer. The treatment paradigm for these patients is rapidly evolving, with future study needed to define the optimal sequencing and potential combinations of these new agents.

This review will examine agents with potential activity in the palliation and treatment of skeletal metastases of prostate cancer, and will weigh the clinical-outcomes evidence for and against their broad use.

Muscle-invasive bladder cancer is an aggressive and potentially lethal disease. Integration of multimodal therapies, improved surgical techniques, and utilization of targeted agents has tremendously improved outcomes.