Multiple Myeloma
Latest News
Latest Videos

CME Content
More News

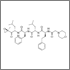
The novel experimental drug sotatercept increased bone mineral density and bone formation in patients with osteolytic lesions of multiple myeloma who had not used bisphosphonates, a phase II study showed.

Results of a new study indicate that half of patients with multiple myeloma were referred to specialist palliative care.

The combination of bortezomib, lenalidomide, and dexamethasone resulted in a partial response or better in more than 60% of patients with relapsed or refractory multiple myeloma, according to results of a phase II trial.

Results of a large meta-analysis indicated that treatment with lenalidomide for newly diagnosed multiple myeloma was associated with an increased risk for developing secondary hematologic malignancies.
Results from a phase I/II trial indicate that carfilzomib may be a safe and effective substitute for bortezomib in multiple myeloma patients whose disease progressed during treatment with a bortezomib-containing regimen.

More than one-third of African American patients with monoclonal gammopathy of undetermined significance or multiple myeloma were found to have an inherited risk factor for the disease, according to the results of a European study.

The International Myeloma Working Group recently released a consensus statement updating recommendations for the management and treatment of patients with multiple myeloma who are not eligible for standard autologous stem-cell transplantation.
Two alternative multiple myeloma plasma cell surface markers have been identified and could be important for subclassification, prognostication, and treatment stratification of patients with multiple myeloma.

The adverse event profile of single-agent carfilzomib suggests that the agent is an important treatment option for patients with advanced multiple myeloma, particularly those with pre-existing peripheral neuropathy or those who are at risk for the development of peripheral neuropathy.

This article reviews the hematologic safety profile of carfilzomib in patients with relapsed/refractory multiple myeloma, as assessed in a cross-trial safety analysis of four phase II studies, and makes recommendations for the appropriate management of hematologic adverse events.
Patients able to achieve stringent complete response after undergoing autologous stem-cell transplantation for multiple myeloma achieved improved long-term outcomes, including overall survival and time to progression, compared with patients who achieved lesser levels of response.
The results of a univariate analysis have shown that the presence of hyperdiploidy and chromosomal aberrations del(17p13), t(4;14), and +1q21 are all associated with shorter time to progression from smoldering multiple myeloma to active disease.
Researchers have helped to better define the spectrum of light chain (AL) amyloidosis by identifying that AL amyloidosis patients with myeloma who have more than 10% bone marrow plasma cells or those with hypercalcemia, renal failure, anemia, and lytic bone lesions have a similar prognosis to patients with overt myeloma.
Adding vorinostat to bortezomib for the treatment of relapsed multiple myeloma resulted in a statistically significant increase in progression-free survival compared with bortezomib alone, according to results of a new study.

The addition of the oral pan-deacetylase inhibitor panobinostat to bortezomib helped to elicit responses in about one-third of heavily pre-treated patients with multiple myeloma who were refractory of bortezomib, according to the phase II results of the PANORAMA 2 study.

Pomalidomide, an immunomodulatory drug, combined with low-dose dexamethasone improved progression-free survival in patients with refractory or relapsed and refractory multiple myeloma compared with standard of care high-dose dexamethasone, according to a new study.

Numerous small series of patients suggest that the prognosis for non-secretory myeloma patients is likely no worse than the prognosis for patients with traditional secretory myeloma, and in some settings may be superior.

Patients with multiple myeloma who underwent autologous stem cell transplantation may have a continued response to the treatment even after the traditional disease assessment at 100 days. A new study indicates that this continued response maintained prognostic value and should be taken into account when considering post-transplant therapies.

Researchers in London have identified a number of new genetic variants that are linked to myeloma, and one specifically linked to a telomerase RNA component gene called TERC, that helps to control the aging process by acting as a cell’s internal clock.
Early treatment with lenalidomide and dexamethasone in patients with high-risk smoldering myeloma significantly delayed progression to symptomatic disease and prolonged survival with a good safety profile.

Older multiple myeloma patients exposed to novel agents prior to autologous peripheral blood stem cell transplantation were at increased risk for engraftment syndrome.

The International Myeloma Working Group recently released new recommendations to aid physicians in the treatment of bone disease related to multiple myeloma.
Results from a first-in-human trial of daratumumab indicate that the investigational drug reduced paraprotein and bone marrow plasma cells at doses greater than 4 mg/kg in patients with advanced multiple myeloma.

Pomalidomide in combination with low-dose dexamethasone had a highly significant benefit on progression-free survival and overall survival compared with single-agent high-dose dexamethasone in patients with relapsed or refractory multiple myeloma, according to updated results of the MM-03 trial presented at the ASCO 2013 Annual Meeting.

Multiparameter flow cytometry and deep sequencing were both able to accurately identify patients with multiple myeloma who were negative for minimal residual disease, a factor that was found to better predict prolonged survival compared with complete response as measured by traditional response criteria.