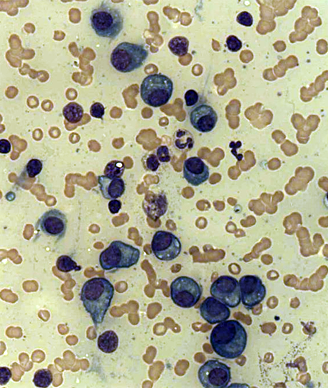
Multiple Myeloma
Latest News
Latest Videos

CME Content
More News

Use of a VD regimen alone to treat patients with transplant-ineligible multiple myeloma was not inferior to VTD or VMP regimens, according to the UPFRONT trial.

Zoledronic acid did not have any antitumor effect in asymptomatic multiple myeloma patients in relapse, but did delay symptomatic progression and bone disease.

Treatment with lenalidomide and the HDAC inhibitor vorinostat may be effective as a maintenance therapy in multiple myeloma patients after autologous transplant.

Autologous stem cell transplant led to significantly longer overall survival than non-transplant strategies in a cohort of elderly patients with multiple myeloma.
The addition of elotuzumab to lenalidomide and dexamethasone improved progression-free survival and overall response rate in relapsed multiple myeloma patients.
Treatment with carfilzomib and dexamethasone doubled the PFS in relapsed multiple myeloma patients compared with treatment with bortezomib and dexamethasone.
Multiple myeloma patients had better overall survival if they had prior knowledge of having monoclonal gammopathy of undetermined significance.

The FDA has approved panobinostat (Farydak), in combination with bortezomib and dexamethasone, for treating patients with multiple myeloma.

The FDA has expanded the use of oral lenalidomide in multiple myeloma to include its use in combination with dexamethasone for newly diagnosed patients.

A scoring system that measured frailty in elderly patients with multiple myeloma was able to accurately predict mortality and risk of toxicity.
The International Myeloma Working Group recently published a series of consensus statements discussing the use of MRI in the treatment of multiple myeloma.
A new trial reported improved overall survival in myeloma patients who received a higher bortezomib dose as part of a bortezomib/melphalan/prednisone regimen.

Cigarette smoking is not a risk factor for multiple myeloma, according to the results of a pooled analysis of nine case-control studies.

The addition of daratumumab to traditional therapies used in the treatment of multiple myeloma was well tolerated and associated with high rates of responses.

Early trial data support further evaluation of ixazomib combined with lenalidomide and dexamethasone for the treatment of newly diagnosed multiple myeloma.

Survivors of multiple myeloma and pancreatic cancer may have some of the poorest physical health-related quality of life outcomes, according to a new study.

Patients who experienced early relapse of multiple myeloma after undergoing autologous stem cell transplantation had worse progression-free and overall survival.

Myeloma patients treated with melphalan prior to transplant had reductions in chemo-induced nausea/vomiting when given granisetron/dexamethasone and aprepitant.
Panobinostat added to bortezomib/dexamethasone for treating relapsed/refractory multiple myeloma improved progression-free survival and complete response rates.

Multiple myeloma patients may already suffer from sensory deficits prior to treatment, likely due to disease-related decreases in peripheral innervation density.
The optimal treatment strategy for newly diagnosed multiple myeloma is consolidation with melphalan, stem-cell transplantation, then lenalidomide maintenance.

Continuous treatment with lenalidomide plus dexamethasone improved progression-free and overall survival in transplant-ineligible multiple myeloma patients.

Treatment with bortezomib prior to autologous stem cell transplant resulted in superior outcomes in multiple myeloma patients with end-stage renal failure.

In a phase I trial, the oral AKT inhibitor afuresertib demonstrated single-agent activity against hematologic malignancies, specifically multiple myeloma.

Researchers from Japan believe they have identified a novel staging system based on hemoglobin and plasmacytoma that may help to stratify patients with multiple myeloma who are treated with novel therapeutics.