
Updates on treatments like vusolimogene oderparepvec for advanced melanoma, and novel therapies have been provided by the FDA.

Ariana Pelosci, managing editor for CancerNetwork® and the journal ONCOLOGY®, has been with the team since June 2021. She specializes in both web and print, and runs the social media accounts for CancerNetwork®.
She graduated from the University of Delaware, where she studied Media Communications and minored in journalism and marketing. At heart, she is a Jersey girl, and you can always find her down the shore during her free time.
Ariana loves to read, specifically historical or contemporary fiction. Follow Ariana on Twitter @APelosci or email her at apelosci@mjhlifesciences.com.

Updates on treatments like vusolimogene oderparepvec for advanced melanoma, and novel therapies have been provided by the FDA.

Only 2 patients died in the phase 3 trial evaluating interferon-alpha in adult patients with cancer, but were deemed unrelated to study treatment.

Pediatric patients self-reported symptom burden using the SSPedi tool, which observed symptom burden from baseline, 4 weeks, and 8 weeks.

Boris M. Kiselev, MD, discusses the role of psycho-oncology in comprehensive cancer care, addressing barriers and offering a vision for the field’s future.

Results from a phase 3 trial led to the approval of leuprolide mesylate as a 3-month formulation for patients with advanced prostate cancer.

Results from the monarchE study showed a statistically significant OS improvement with abemaciclib plus ET for patients with HR+/HER2– early breast cancer.
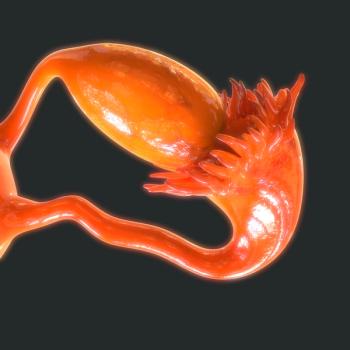
The novel treatment was evaluated in a phase 2 trial and saw a clinical benefit in patients with advanced ovarian cancer.

Declan Walsh, MD, highlighted how the supportive and palliative care space has evolved since he first began working in the field.

Results from the phase 3 VERIFY trial of rusfertide for erythrocytosis in patients with polycythemia vera led to the FDA decision.
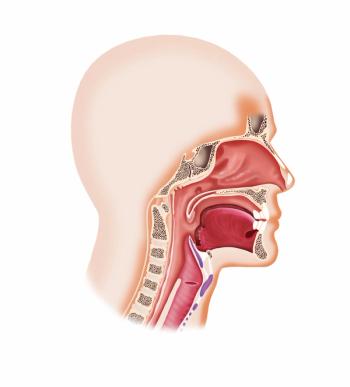
The Chemo Mouthpiece had favorable outcomes when used during and after treatment with chemotherapy among a range of patients with various types of cancer.

The confirmed ORR was 40.0% in cohort 1A and 66.7% in cohort 1B of patients treated with sacituzumab tirumotecan/tagitanlimab for advanced NSCLC.

A survey found that patients believed dermatologic AEs were more prevalent with anti-cancer therapies than what has been reported.

Tran Ho, DO, FSSO, FACS, discussed how she found her passion in breast surgical oncology and how her mentors helped her achieve success.

Balancing a career as a pediatric oncologist with leadership responsibilities, Maria C. Velez, MD, shares insights into her passion for the field and ASPHO’s impactful work.

Data from the LITESPARK-005 Q-TWiST analysis show belzutifan’s improved quality-adjusted survival time and favorable toxicity profile vs everolimus in RCC.

Priya Jayachandran, MD, FACP, spoke about her new position at El Camino Health as well as her career as a breast oncologist.

Bhuvanesh Singh, MD, has big plans for enhancing the skin cancer program at Northwell Health.

Patients with FRα+, platinum-resistant high-grade serous epithelial ovarian, fallopian tube, or primary peritoneal cancer may now be eligible for mirvetuximab soravtansine in the UK.

Results from the AQUILA trial led to the approval of subcutaneous daratumumab for patients with smoldering multiple myeloma at risk of developing the disease.

Updated POSITIVE trial findings showed no significant breast cancer events in patients who breastfed following endocrine therapy for HR+ breast cancer.

Phase 2b SunRISe-1 trial findings supported the FDA to grant priority review to TAR-200 in BCG-unresponsive high-risk NMIBC with carcinoma in situ.

Paolo Tarantino, MD, PhD, discusses sequencing ADCs, managing cross-resistance, and understanding ADC composition for patients with breast cancer.

Heather McArthur, MD, focused on current and future strategies for treating early-stage breast cancer.
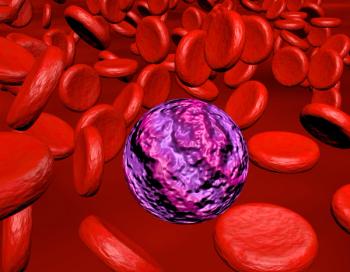
A PDUFA date for decitabine/cedazuridine and venetoclax in newly diagnosed AML has been set for February 25, 2026.

SIR-Sphere Y-90 resin microspheres are the only approved radioembolization treatments in the US for both HCC and mCRC.

Updated guidelines from ASTRO recommend various radiation therapy techniques for patients with WHO grade 4 adult-type diffuse glioma.

Findings from a WeCanWork study showed that men who work laborious jobs need additional support during and after cancer treatment.

The EPCORE NHL-1 trial showed a 41% complete response rate with epcoritamab for patients with relapsed/refractory LBCL.

Nicholas J. Hornstein, MD, PhD, highlighted results from the phase 3 ATOMIC and CHALLENGE trials in CRC presented at the 2025 ASCO Annual Meeting.

The Adventist Health Study results found that patients who were vegetarians had a lower risk of medium-frequency cancers.