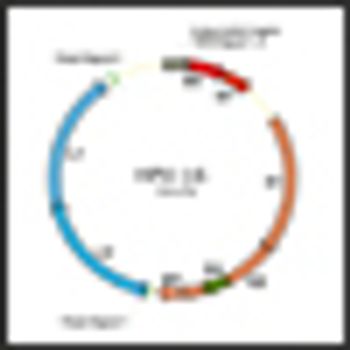
A study published in Lancet Oncology shows that an AS04-adjuvanted HPV 16 and HPV 18 vaccine developed by GlaxoSmithKline Biologicals can offer protection against anal cancer.

A study published in Lancet Oncology shows that an AS04-adjuvanted HPV 16 and HPV 18 vaccine developed by GlaxoSmithKline Biologicals can offer protection against anal cancer.
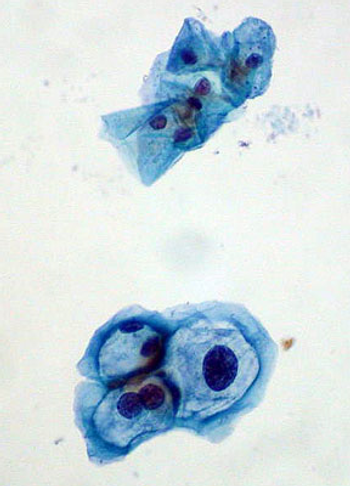
One of the highlights of the released abstracts is “Cervical cancer risk for 330,000 women undergoing concurrent HPV testing and cervical cytology in routine clinical practice” (J Clin Oncol 29: 2011 (suppl; abstr 1508). The large-scale study showed the effectiveness of human papillomavirus (HPV) testing alone or in combination with cytology testing for identifying women at high-risk for cervical cancer development.

Human papillomavirus (HPV) is the most common sexually transmitted infection in the United States, with approximately 20 million people currently infected and an additional 6.2 million infected each year, despite increased media attention to HPV as a cause of cervical cancer and the availability of a vaccination to reduce HPV-associated cervical cancer.

Investigators from the German Cancer Research Center in Heidelberg reported on the effects of LMV-601 on cultured human cervical cells. LMV-601 is a phosphatidylcholine-specific phospholipase C (PC-PLC) inhibitor.

With the increased use of human papillomavirus vaccines such as Gardasil and Cervarix, the medical community is likely to see a decrease in cases of genital warts and other complications caused by several HPV strains. But it may be a decade or two before oncologists can expect to see a decline in cervical cancer rates attributable to the use of these relatively new vaccines.

Human papilloma virus often lurks in cervical tissue, and it can cause cancer there. But the infection is also often benign, particularly among young women. Biomarkers of transformation are proving useful in helping cytologists to decide when a suspicious-looking Pap result is truly a sign of trouble.

Of the predominant gynecologic cancers, cancer of the uterine cervix is the least common, with only 11,270 new cases anticipated in the United States in 2009. Nevertheless, approximately 4,070 women die of cancer of the uterine cervix annually in the United States.

The American College of Obstetricians and Gynecologists has recommended that women begin cervical cancer screening at age 21 rather than three years after the onset of sexual activity, as was previously recommended by the group.

Out with the old and in with the new is a commonly followed maxim in medicine given the rapid pace of developments in diagnosis and treatment. Human papillomavirus vaccines are relative newcomers to the cervical cancer armamentarium, but they cannot be relied on to do the job on their own; screening is still a must.

Cervarix has won FDA approval for the prevention of cervical pre-cancers and cervical cancer associated with HPV-16 and HPV-18 for use in girls and young women (ages 10-25), according to GlaxoSmithKline.

PET/CT is gaining recognition both medically and politically when it comes to sizing up cancer. And the timing for oncologists couldn’t be better.

The US Food and Drug Administration (FDA) has approved GlaxoSmithKline’s human papillomavirus bivalent (types 16 and 18) vaccine, recombinant (Cervarix) for the prevention of cervical precancers and cervical cancer associated with oncogenic human papillomavirus (HPV) types 16 and 18 for use in girls and young women (aged 10–25).

The Society of Gynecologic Oncologists (SGO) has published the first in a series of four papers on a variety of cervical cancer issues and topics that were the focus of its Forum “The Future Strategies for Cervical Cancer Prevention: What Do We Need to Do Now to Prepare,” held last September in Chicago.

The final analysis of the largest efficacy trial of a cervical cancer vaccine was published July 25, 2009, in The Lancet. The study, involving 18,644 women, confirmed GlaxoSmithKline’s Cervarix is highly effective at protecting against the two most common cervical cancer–causing human papillomavirus (HPV) types, 16 and 18. The study also showed that the vaccine provides cross-protection against HPV types 31, 33, and 45, the three most common cancer-causing virus types beyond 16 and 18.

Imaging with MR and/or PET/CT can help physicians figure out appropriate treatment for cervical cancer patients as well as prevent unnecessary therapy, according to a new study.

Results from an 8-year trial involving more than 130,000 women published in The New England Journal of Medicine (360:1385-1394, 2009) demonstrate that in low-resource settings, a single round of human papillomavirus (HPV) testing significantly reduces the numbers of advanced cervical cancers and deaths, compared with Pap testing or visual inspection with acetic acid (VIA). This was the first randomized controlled trial to measure incidence of cervical cancer and associated rates of death as the primary outcomes, using different tools for screening.

Diffusion-weighted MRI added to standard T2-weighted scans can help spot cervical cancer in its early stages. A preliminary study from the Institute of Cancer Research in London determined that DWI can spot tumors missed by T2 imaging and bolster management options for women who wish to preserve reproductive organs.

A proposal to tighten up cervical cancer proficiency testing should lead to a decrease in the number of false-negative cases that cross the desks of gynecologic oncologists.

BD Diagnostics recently announced that it received US Food and Drug Administration (FDA) Premarket Approval for the BD FocalPoint GS Imaging System.

Merck & Co recently announced that the US Food and Drug Administration (FDA) has accepted, and designated for priority review, the supplemental Biologics License Application (sBLA) for its recombinant human papillomavirus quadrivalent vaccine (Gardasil) for potential use in women aged 27 through 45.

The cytoprotective agent amifostine (Ethyol) did not reduce the rate of acute severe toxicity among patients undergoing combined radiation therapy and chemotherapy for locally advanced cervical cancer, according to a phase I/II study reported at ASTRO 2007 (abstract 10). The trial, RTOG 0116, enrolled women with cervical cancer who had evidence of positive para-aortic or high common iliac nodes.

Phase III data showed that at 18 months after the first of a three-dose regimen, 100% of women up to age 55 vaccinated with the GlaxoSmithKline (GSK) cervical cancer candidate vaccine (Cervarix) had antibodies present against the two most common cancer-causing human papillomavirus types, 16 and 18

Merck & Co., Inc. has committed to donate at least 3 million doses of quadrivalent human papillomavirus (HPV types 6, 11, 16, 18) recombinant vaccine (Gardasil), the cervical cancer vaccine, for use in demonstration projects in lowest-income nations throughout the world.

Merck to donate 3 million doses of Gardasil to poor countries

Vaccines have been exceptionally effective against diseases such as smallpox, measles, chickenpox, and polio. They are among the safest and most cost-effective agents for disease prevention. In recent years, vaccination has been considered for other diseases, including AIDS and cancer. Cancer vaccines can be categorized as preventive or therapeutic. Preventive vaccines, which are commercially available for cervical cancer and liver cancer, block infection with the causative agents of human papillomavirus and hepatitis B virus, respectively. The benefit of cancer treatment vaccines lies in their ability to "boost" the immune system response to cancer cells, which is generally low. Using vaccines in the treatment of cancer is relatively new, however, and chiefly experimental. Therapeutic vaccines for breast, lung, colon, skin, renal, prostate, and other cancers are now being investigated in clinical trials. Oncology nurses may play a significant role in reducing barriers to uptake of preventive vaccines among the general public and in increasing patients' acceptance of therapeutic cancer vaccines.