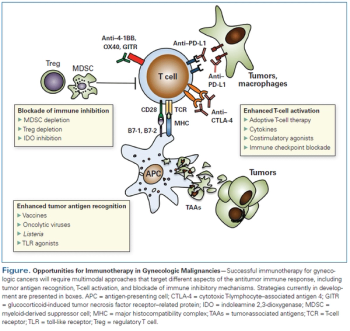
A large cohort study found no concerning safety issues associated with the use of human papillomavirus (HPV) vaccine in adult women. There was an increased rate of celiac disease, but this may be related to general underdiagnosis of the condition and its unmasking at vaccination visits.