
However, researchers did identify a positive correlation for the risk of some cancers in this prospective cohort study.
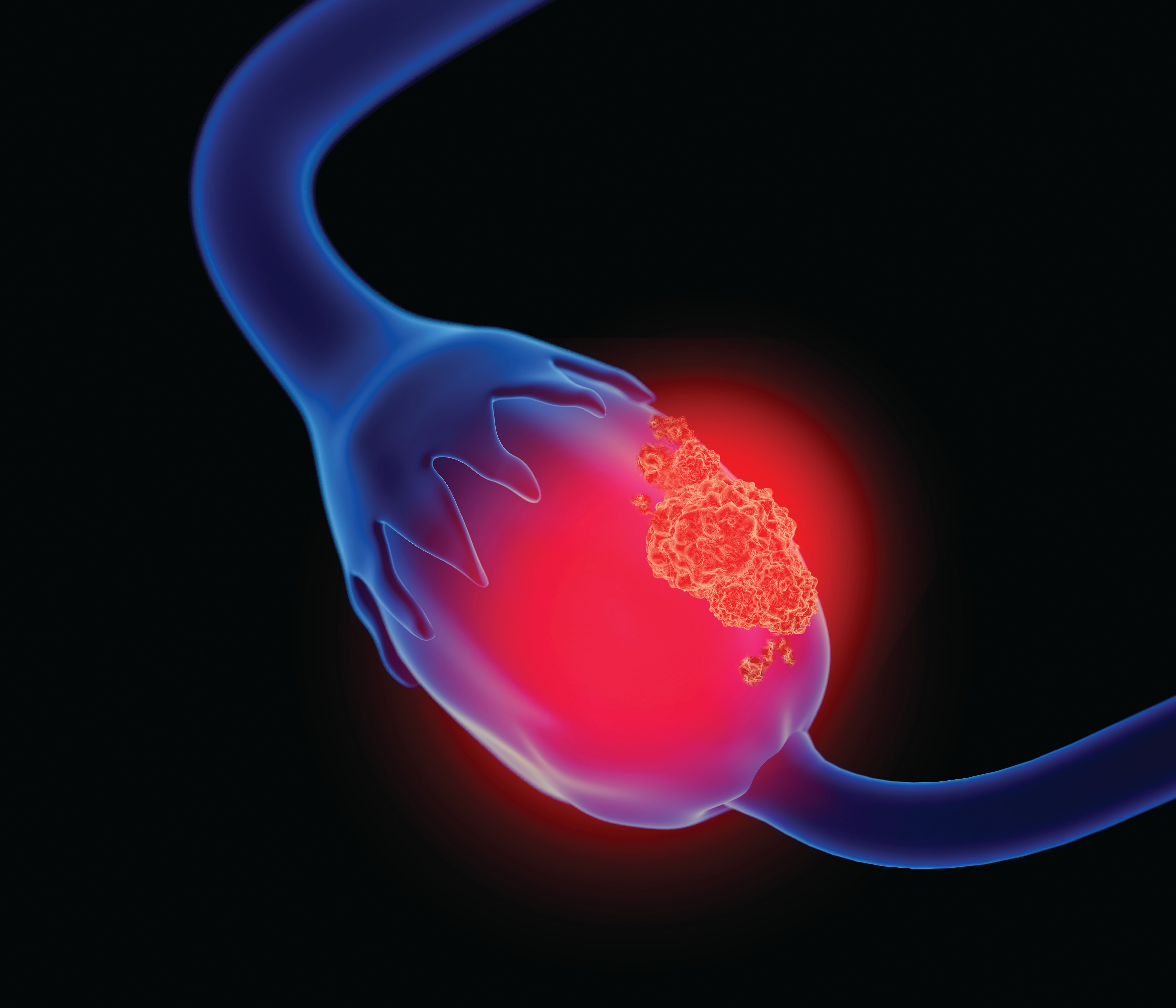
Atezolizumab Plus Bevacizumab/Chemo Does Not Significantly Improve PFS for Ovarian Cancer

However, researchers did identify a positive correlation for the risk of some cancers in this prospective cohort study.
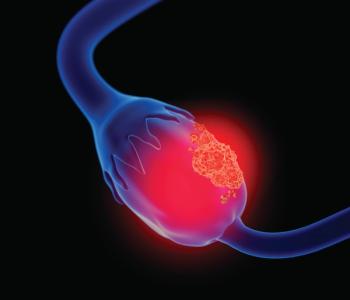
The recommendations were based on a review of 17 clinical trials, FDA approvals, and consensus where evidence was lacking.

The US Oncology expert looks back on how far the ovarian cancer treatment space has come, and where it is headed.
The expert from Dana-Farber Cancer Institute spoke about the study of pembrolizumab monotherapy in patients with advanced recurrent ovarian cancer.

The US Oncology expert noted that input of the multidisciplinary board is a great place to air out concerns regarding ovarian cancer treatment.
This study identified specific variants in multiple Papanicolaou tests from the same patients conducted up to 6 years before the diagnosis of high-grade serous epithelial ovarian cancer.

The expert from the Dana-Farber Cancer Institute discussed the study findings which assessed the use of pembrolizumab monotherapy in patients with advanced recurrent ovarian cancer.

The US Oncology expert explained that results from 2 trials presented at the 2020 ASCO Virtual Program may change how oncologists choose to treat these patients.

The US Oncology expert reported that secondary surgical cytoreduction may not lead to improved outcomes in women platinum-sensitive, recurrent ovarian cancer.

The MSK expert discussed how pre- and post-genetic testing online education made patients more knowledgeable about genetic testing.
These results suggest the need for future studies to examine scalable interventions to improve fatigue in women with gynecologic cancers.

The MD Anderson Cancer Center expert discussed further research in delivering online accessible genetic testing.

The expert from Dana-Farber Cancer Institute discussed the study design and inclusion criteria for this study of pembrolizumab in patients with advanced recurrent ovarian cancer.

The MD Anderson Cancer Center expert spoke about the key takeaways from a trial designed to test whether pre and/or post-test genetic counseling is needed to optimally deliver online accessible genetic testing.

The expert in ovarian cancer indicated that treatment with the studied maintenance olaparib tablets could aid patients in having a more regular lifestyle.
The study showed that the addition of atezolizumab to bevacizumab, paclitaxel, and carboplatin as a front-line treatment in women with newly diagnosed advanced stage ovarian cancer did not meet its primary endpoint of progression-free survival.

The expert discussed study methods from the NRG Oncology/Gynecologic Oncology Group (GOG) study presented at the 2020 ASCO Virtual Program.

At the 2020 ASCO Virtual Scientific Program, Karen H. Lu, MD, presented on findings from the MAGENTA trial, designed to test whether pre and/or post-test genetic counseling is needed to optimally deliver online accessible genetic testing.

The expert discussed study methods and results from the MAGENTA trial evaluating pre and/or post-test genetic counseling.

The results of the trial demonstrated that maintenance olaparib provided an unprecedented improvement in median OS versus placebo in patients with platinum-sensitive, relapsed ovarian cancer and a BRCA mutation.
A study presented at the AACR Virtual Annual Meeting II showed a significant decrease in mortality rate for patients with epithelial ovarian cancer when treated with lipophilic statins, supporting the further evaluation of statins to treat ovarian cancer in randomized clinical trials.

The Dana-Farber Cancer Institute expert discussed pembrolizumab monotherapy in patients with advanced recurrent ovarian cancer.
The combination use of niraparib plus bevacizumab significantly improved clinical outcomes, compared with niraparib alone, in patients with recurrent ovarian cancer.
The MD Anderson Cancer Center expert spoke about the disconnect in women who are at risk for ovarian cancer to actually receive genetic counseling.
Recent findings suggested that watching Disney movies during chemotherapy treatment may be correlated with improvements in quality of life in patients with gynecologic cancers.