
Treatment with liso-cel appears to result in clinically meaningful activity across several subgroups of patients with mantle cell lymphoma, including those with high-risk features.

Your AI-Trained Oncology Knowledge Connection!


Hayley Virgil is a senior editor with CancerNetwork. When she isn't traveling to conferences and championing health equity in the oncology space, she can be found hiking, foraging wild plants, gardening, sewing ballgowns, practicing embroidery, or playing video games.

Treatment with liso-cel appears to result in clinically meaningful activity across several subgroups of patients with mantle cell lymphoma, including those with high-risk features.
Patients with benign thyroid tumors resected with a transoral endoscopic thyroidectomy vestibular approach have less complications vs those treated with a standard surgical approach.
The FDA previously granted approval to frontline olaparib plus abiraterone/prednisone in May 2023 for BRCA1/2–, DDR–altered metastatic castration-resistant prostate cancer.

Patients with metastatic cancer who are receiving treatment at nonacademic or low-volume facilities appear more likely to receive end of life immunotherapy.

Cancer incidence appears to have increased from 2015 to 2019 across several disease types, including breast, pancreatic, and uterine cancers.

Data from the phase 3 LUNAR trial support the use of tumor treating fields in patients with non–small cell lung cancer.

Investigators indicate that ongoing efforts should focus on meeting the needs of adult cancer survivors with co-morbid substance use disorder, with an emphasis on prioritizing populations in which the disorder is highly present.
Investigators also report that patritumab deruxtecan is well tolerated in those with EGFR-mutant non–small cell lung cancer in the HERTHENA-Lung01 study.

Investigators report favorable overall survival among patients with chronic lymphocytic leukemia who receive ibrutinib/venetoclax over FCR.
Investigators note that the phase 3 VESPER study’s data support the use of 6 neoadjuvant cycles of dose-dense MVAC vs 4 in muscle-invasive bladder cancer.
177Lu-PNT2003 is a generic version of lutetium Lu 177 dotatate, which is used to treat somatostatin receptor–positive gastroenteropancreatic neuroendocrine tumors.
Plans have been put in place to launch a phase 1/2 study assessing NUV-1511 in a population of patients with advanced solid tumors.
Data demonstrate efficacy of robotic surgery for colectomies, which is swiftly becoming the 'preferred approach' for surgeons.

Patients with relapsed/refractory acute myeloid leukemia appear to benefit from treatment with SLS009.
![[18F]TFB PET/CT May Predict [18F]TFB/FDG Positivity in Differentiated Thyroid Cancer | Image Credit: © Kateryna Kon - shutterstock.com](https://cdn.sanity.io/images/0vv8moc6/cancernetwork/b4ee57eff4c36f6c8244904cf57aa642e4da812c-1000x667.jpg?w=350&fit=crop&auto=format)
The predictive value of [18F]TFB PET/CT may be useful in identifying patients with differentiated thyroid cancer who may be eligible for treatment with [131I]iodine therapy.
Data from the phase 2 A-PLUS trial indicate that bevacizumab plus chemotherapy prior to whole brain radiotherapy may be an efficacious systemic strategy for intractable brain and extracranial metastases stemming from breast cancer.

Investigators report that prediabetes may prove to be a target for intervention to decrease morbidity and mortality in survivors of pediatric cancer.

Investigators note that the benefit of maintenance olaparib rechallenge in previously treated, platinum-sensitive ovarian cancer extends to the BRCA-mutant and non-mutant cohorts.

Results from a long-term analysis of the phase 3 IMCgp100-202 trial indicate that tebentafusp results in better disease control and long-lasting responses in those with HLA-A*02:01–positive, previously untreated metastatic uveal melanoma.
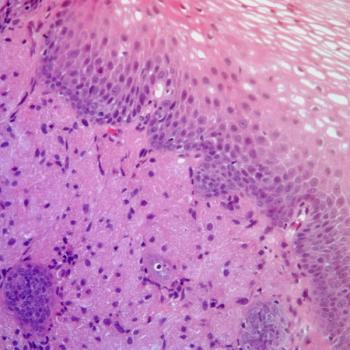
Sugemalimab is now approved for managing esophageal squamous cell carcinoma in China following results from the phase 3 GEMSTONE-304 study.
The application for amivantamab and lazertinib for locally advanced or metastatic non–small cell lung cancer is supported by data from the phase 3 MARIPOSA trial.
Investigators indicate that, although treatment with adjuvant/neoadjuvant pembrolizumab results in an absolute decrease in risk of an event, the benefit was not statistically significant in those with untreated, locally advanced gastric/GEJ cancer.
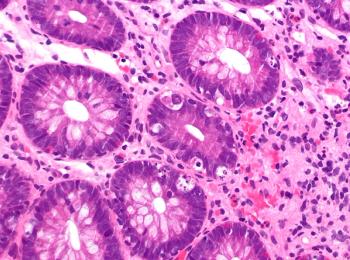
Belumosudil is the first therapy within England and Wales available for the treatment of patients 12 years and older who have previously undergone a minimum of 2 lines of systemic treatment.

Lower risk of disease progression following treatment with atezolizumab and carboplatin for metastatic triple-negative breast cancer appears to be associated with increases in tumor infiltrating lymphocytes and tumor mutational burden.
Minimal residual disease testing appears to be a valuable tool for predicting outcomes and potential relapse in patients with lymphoid malignancies.
Data from part 2 of the phase 3 RUBY trial support the use of dostarlimab as a backbone for immunotherapy-based combinations in patients with primary advanced or recurrent endometrial cancer.

In Europe, pembrolizumab is now available as a treatment in combination with fluoropyrimidine-/platinum-containing chemotherapy for HER2-negative gastric or GEJ adenocarcinoma, as well as in combination with gemcitabine/cisplatin for locally advanced biliary tract carcinoma.
mRNA-4157 is being evaluated in combination with pembrolizumab as part of the phase 2b KEYNOTE-942/mRNA-4157-P201 trial in patients with completely resected, high-risk, stage III to IV melanoma.
A regimen consisting of venetoclax, ibrutinib, prednisone, obinutuzumab, and lenalidomide appears safe for a population of patients diagnosed with mantle cell lymphoma, as investigators report no significant dose-limiting toxicities or tumor lysis syndrome.

Adding camrelizumab to chemotherapy appears tolerable for patients with resectable stage IIIA or IIIB non–small cell lung cancer in the phase 2 TD-FOREKNOW trial.
September 30th 2023