
About a fifth of patients with lung cancer in the United States receive no treatment at all, while approximately 6 in 10 receive the minimal treatments recommended by the National Comprehensive Cancer Network.

Your AI-Trained Oncology Knowledge Connection!


About a fifth of patients with lung cancer in the United States receive no treatment at all, while approximately 6 in 10 receive the minimal treatments recommended by the National Comprehensive Cancer Network.
The University of Texas MD Anderson Cancer Center and Takeda Pharmaceutical Company Limited have entered an exclusive license agreement and research agreement to develop and market chimeric antigen receptor-directed natural killer-cell therapies.
Proton therapy’s direct and precise application of radiation for mid-stage lung cancer is being investigated in an ongoing phase III trial at the University of Cincinnati.
A blood test which may detect the traces of developing breast cancer through autoantibodies years before clinical signs show themselves is now proposed by researchers in the United Kingdom.

A new study in the Journal of Clinical Oncology finds that a subset of pediatric ALL relapses may be triggered by the very chemotherapy which helped patients beat the cancer the first time.
Conversations about the financial realities of genomic testing are not always taking place, according to a new study by the American Cancer Society.

Treatment with lenvatinib and pembrolizumab has shown promise in patients with metastatic clear cell renal cell carcinoma who have already received an immune checkpoint inhibitor.
Massachusetts General Hospital’s Severe Immunotherapy Complications Service focuses on the increasing number of “rare” cases of immunotherapy-related adverse events.
A new investigational drug called ADCT-402 that combines a cell-killing compound with a monoclonal antibody shows early promise for patients with diffuse large B-cell lymphoma.
Machine learning of ultrasound scans could provide a preliminary method of screening for thyroid cancer.

The probiotics and prebiotics of yogurt and fiber consumption is associated with decreased risk of lung cancer.
Endometrial cancer survival correlates with total number of chemotherapy cycles – but not whether they are started earlier than radiation, according to the results of a new study.
A compound called MRTX849 is showing signs of being the first effective inhibitor of the KRAS G12C-mutant subtype of non-small cell lung and colorectal cancers.
Using RNA sequencing for individual pediatric cancer cases could more effectively target gene expression in tumors.
A new inhibitor may help patients with a certain subset of head and neck squamous cell carcinoma.
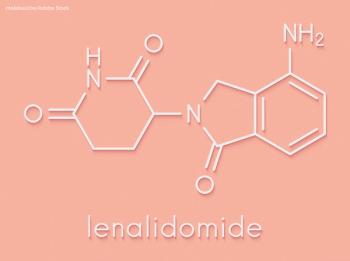
Early treatment with lenalidomide can stave off multiple myeloma and provide longer progression-free survival (PFS) than just monitoring alone.

A new machine-learning computer model helped predict which patients of cancer may die soon.
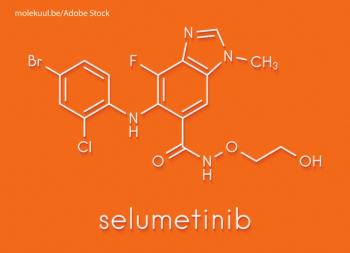
The MEK inhibitor selumetinib showed clinical responses in the plexiform neurofibromas of adult patients with neurofibromatosis type 1.

Patients, providers, and healthcare processes benefit from systematically collecting patient-reported outcomes and reviews of their experiences in breast cancer treatment.

The worldwide incidence, and death toll, of colorectal and pancreatic cancers have sharply increased since 1990, according to the results of a new study.
The epigenetic signals in circulating cell-free DNA (cfDNA) in patients diagnosed with diffuse large B-cell lymphoma (DLBCL) could predict which cases are the most likely to recur.

Conversion of tryptophan to kynurenine was associated with lower survival rates.

Immunotherapy responses may not be equal between the sexes.

The approval comes one day after the FDA approved niraparib for the same subset of ovarian cancer.
Tucatinib showed improvements in survival in patients with HER2-positive breast cancer.
The results of this new study could also further explain the patterns of tumor development among certain ethnicities.

Alcohol has been labeled a carcinogen by a growing number of agencies across the globe, after years of scientific and policy debate. But just how much of the cancer burden is it responsible for?
Radiologists and data scientists decided to get together to evaluate the effectiveness of artificial intelligence and human observation when it comes to detecting breast cancer in mammograms.

Niraparib was approved by the (FDA) for previously treated advanced ovarian, fallopian, or primary peritoneal cancers.

Published: September 19th 2019 | Updated:
Published: September 23rd 2019 | Updated:
Published: September 24th 2019 | Updated:
Published: September 25th 2019 | Updated:
Published: September 26th 2019 | Updated:

Published: September 26th 2019 | Updated: