The CAR T-cell therapy axicabtagene ciloleucel induced a median overall survival of 25.8 months for patients with refractory large B-cell lymphoma.

The CAR T-cell therapy axicabtagene ciloleucel induced a median overall survival of 25.8 months for patients with refractory large B-cell lymphoma.

The FDA accepted a supplemental biologics license application and granted a priority review to durvalumab for the treatment of patients with previously untreated extensive-stage small cell lung cancer.

The FDA has granted priority review to a supplemental biologics license application for pembrolizumab for the treatment of patients with BCG-unresponsive, high-risk, non-muscle invasive bladder cancer.

Robert A. Brodsky, MD discussed the study of a first-of-kind multi-antigen targeted off-the-shelf chimeric antigen receptor- natural killer cell therapy with engineered persistence that will be presented at the ASH Annual Meeting & Exposition.
The MSK expert discusses exciting advancements in the field of immunotherapy for treating lung cancer at the Annual New York Lung Cancers Symposium®.
Beth Eaby-Sandy, a nurse practitioner, discussed AEs associated with immunotherapy treatment in patients with lung cancer.
Marjorie Zauderer, MD, from Memorial Sloan Kettering Cancer Center, discussed new immunotherapy approaches at the 34th Annual Meeting & Pre-Conference Programs of the Society for Immunotherapy of Cancer (SITC 2019).
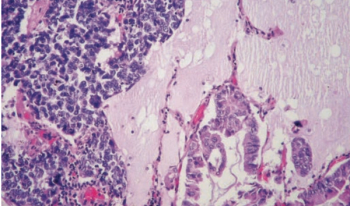
ONCOLOGY recently spoke with Dr. Sandip Patel about a potential novel therapeutic ap- proach to treating patients with neuroendocrine tumors.
Domenico Mallardo, MD, Istituto Nazionale Tumori "Fondazione Pascale" in Naples, discusses results from his trial – designed to evaluate anti-CTLA4 agents in patients with melanoma who relapsed on treatment with ipilimumab (Yervoy), which was presented at the 34th Annual Meeting & Pre-Conference Programs of the Society for Immunotherapy of Cancer (SITC 2019).

Luca Biavati, MD, from Johns Hopkins Medicine, discussed bone marrow T-cells and bone marrow infiltrating lymphocytes as a source for adoptive cell therapy at the 34th Annual Meeting & Pre-Conference Programs of the Society for Immunotherapy of Cancer (SITC 2019).

Robert Ferris, MD, from UPMC Hillman Cancer Center, discusses the 34th Annual Meeting & Pre-Conference Programs of the Society for Immunotherapy of Cancer (SITC 2019), and how immunotherapy will continue to transform cancer treatment.

Jason Williams, MD, discusses the development of intratumoral immunotherapy, as well as trial results from his poster presentation at the 34th Annual Meeting & Pre-Conference Programs of the Society for Immunotherapy of Cancer (SITC 2019).

Joshua Brody, MD, discusses the gratification of the crossover between medicine and science at the 34th Annual Meeting & Pre-Conference Programs of the Society for Immunotherapy of Cancer (SITC 2019).

Mario Sznol, MD, from Yale University, discusses where the field of immune-oncology will be 1 and 5 years from now at the 34th Annual Meeting & Pre-Conference Programs of the Society for Immunotherapy of Cancer (SITC 2019).
The University of Texas MD Anderson Cancer Center and Takeda Pharmaceutical Company Limited have entered an exclusive license agreement and research agreement to develop and market chimeric antigen receptor-directed natural killer-cell therapies.
A compound called MRTX849 is showing signs of being the first effective inhibitor of the KRAS G12C-mutant subtype of non-small cell lung and colorectal cancers.
A new inhibitor may help patients with a certain subset of head and neck squamous cell carcinoma.
A look at the latest lung cancer research, recent advances in the field and their clinical applications.

Immune checkpoint inhibitors have demonstrated impressive activity in patients with CRC and other solid tumors that are dMMR. Researchers explore the data.

This year has seen many advances in immunotherapy. Here is a look at some of the most significant research.
The VOLFI trial enrolled 105 patients with RAS wild-type metastatic colorectal cancer.
Combination therapy with nivolumab and bevacizumab appeared to have clinical activity in ovarian cancer patients with platinum-sensitive or platinum-resistant disease.
A review of recent advances in the noninvasive imaging of immunotherapeutic targets.

Over that time, the number of active drugs in development has grown by more than 90%.
Age disparities in oncology trials are continuing, despite opposition to this issue.