Ovarian Cancer
Latest News
Latest Videos
CME Content
More News

Sharyn Lewin, MD, FACS, shares how she discusses side effects with patients receiving immune checkpoint inhibitors for advanced/recurrent endometrial cancer.

Experts explain what they are looking forward to in the future of treatment for patients with ER/PR+ and P53 wild-type or mutated advanced/recurrent endometrial cancer.

The panel discusses their opinions on the use of immunotherapy in different populations of women with advanced/recurrent endometrial cancer.
Prexasertib is currently under investigation as part of a phase 2 trial as a treatment for patients with platinum-resistant ovarian cancer, endometrial adenocarcinoma, and urothelial cancers.

Based on findings from the phase 3 MIRASOL trial, investigators plan to submit a supplemental biologics license application for mirvetuximab soravtansine in folate receptor α–positive platinum-resistant ovarian cancer.

Patients with platinum-resistant high-grade serous carcinoma have little overall survival benefit with the addition of vistusertib to weekly paclitaxel.
Data from the interim analysis of the phase 3 FLAMES study indicate that senaparib may lengthen progression-free survival in patients with advanced ovarian cancer.
Patients with advanced high-grade epithelial BRCA mutation–negative ovarian cancer appear to benefit from treatment with olaparib and durvalumab plus bevacizumab and chemotherapy.

Patients with resistant or refractory ovarian cancer experience clinical benefits after receiving botensilimab plus balstilimab in the phase 1 C-800 study.

Niraparib maintenance therapy for recurrent ovarian cancer does not yield a significant overall survival benefit in an updated analysis of the phase 3 ENGOT-OV16/NOVA study.

An expert from Dana-Farber Cancer Institute discusses findings from the final overall survival analysis of the phase 3 ENGOT-OV16/NOVA trial.
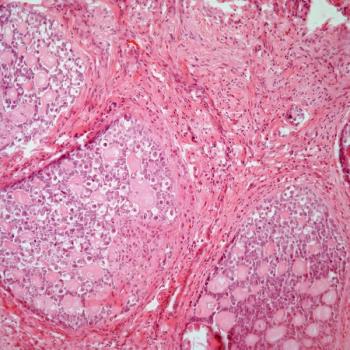
The Carolina Frailty Index Score indicates that patients with ovarian cancer classified as frail are highly more likely to die than patients classified as robust.
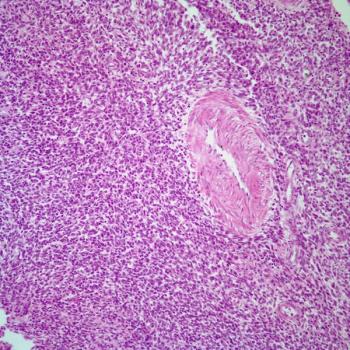
In the SOLAR phase 1b trial, a clinical benefit was observed when patients with RAS-mutated ovarian or endometrial cancer were given olaparib plus selumetinib.

A post hoc analysis demonstrated that dose modifications to frontline niraparib maintenance therapy did not affect progression-free survival among patients with newly diagnosed, advanced ovarian cancer.

The use of palliative care in ovarian cancer resulted in a decrease in overall readmissions and index hospitalization costs.

Durvalumab plus tremelimumab in combination with neoadjuvant chemotherapy did not yield any major adverse effects in patients with newly diagnosed advanced ovarian cancer in the phase 2 KGOG3046 trial.
Minimally invasive surgery for interval debulking resulted in a lower mortality at the 30- and 90-day time points compared with laparotomy in advanced ovarian cancer.
Vadim Gushchin, MD, says that treatment with cytoreductive surgery and hyperthermic intraperitoneal chemotherapy should not be limited by concerns of negatively impacting health-related quality of life in ovarian cancer.

Data from the phase 2 INNATE trial indicate that pimivalimab plus JTX-8064 also appears to be well tolerated in patients with platinum-resistant ovarian cancer.
Maintenance olaparib plus bevacizumab may improve survival in patients with high-grade ovarian cancer regardless of BRCA mutation location, according to a subgroup analysis of the PAOLA-1/ENGOT-ov25 trial.
Treatment with avutometinib and defactinib in patients with low-grade serous ovarian cancer resulted in a positive objective response rate.
Niraparib Maintenance Demonstrates Favorable OS Trend in Platinum-Sensitive Recurrent Ovarian Cancer
Findings from the phase 3 NORA study identified a numerically longer median overall survival among patients with platinum-sensitive recurrent ovarian cancer treated with maintenance niraparib regardless of BRCA mutation status.

The first-in-class Heat Shock Factor 1 pathway inhibitor NXP800, the subject of an ongoing phase 1 dose-escalation study, has been granted a Fast Track designation for the treatment of platinum-resistant ovarian cancer by the FDA.
A longer, 30-month treatment duration with bevacizumab did not improve outcomes over the standard 15-month treatment duration in patients with newly diagnosed ovarian cancer.

Ursula A. Matulonis, MD, spoke about the next steps for mirvetuximab soravtansine in the treatment of patients with folate receptor α–high platinum-resistant ovarian cancer.