
Alisertib yielded promising progression-free survival in advanced/metastatic soft-tissue sarcoma patients, though it did not meet its primary response rate endpoint.

The FDA granted accelerated approval to olaratumab (Lartruvo) in combination with doxorubicin for the treatment of soft-tissue sarcomas that is not amenable to curative treatment with radiotherapy or with surgery and with a histologic subtype treatable with anthracycline-containing regimens.

Alisertib yielded promising progression-free survival in advanced/metastatic soft-tissue sarcoma patients, though it did not meet its primary response rate endpoint.

A comprehensive genomic analysis of more than a thousand sarcoma patients found that about half had putatively pathogenic variations in either known or novel cancer genes.
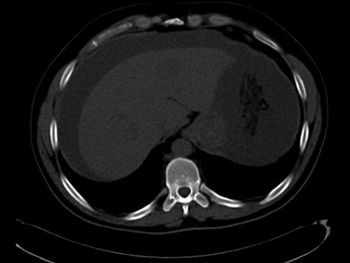
A 37-year-old Lebanese male with no significant past medical history initially presented with an increase in abdominal girth over a few weeks with worsening shortness of breath, nausea, and intermittent vomiting.

Survivors of fusion-negative sarcomas have a higher risk of developing a second malignant neoplasm than those who had fusion-positive sarcomas.

A phase I trial of selinexor found the drug was reasonably well tolerated and showed some activity in patients with advanced soft-tissue or bone sarcoma with progressive disease.

Zoledronate in combination with chemotherapy failed to improve outcomes over chemotherapy alone in patients with osteosarcoma.

Three single-nucleotide polymorphisms were identified that are significantly associated with OS in two separate cohorts of pediatric Ewing sarcoma patients.

A phase II trial showed that the tyrosine kinase inhibitor anlotinib has promising efficacy across a variety of subtypes of advanced refractory soft-tissue sarcoma.

A combination of the endoglin antibody TRC105 and pazopanib shows promising activity in patients with advanced soft-tissue sarcoma, particularly angiosarcoma.

Combined therapy with gemcitabine and pazopanib appears to be a viable treatment option for patients with soft-tissue sarcomas refractory to anthracycline or ifosfamide.

Understanding quality of life and the detrimental impact of disease progression is critical for long-term care and survival of patients with soft-tissue sarcoma.

Busulfan/melphalan high-dose chemotherapy consolidation is associated with better survival vs VAI among patients with localized high-risk Ewing sarcoma.

Regorafenib is associated with improved progression-free survival in pretreated patients with advanced refractory non-liposarcoma soft-tissue sarcomas.

Radiotherapy delivered either pre- or postoperatively improved overall survival compared with surgery alone in patients with retroperitoneal sarcoma.

The addition of trabectedin to doxorubicin was not superior to doxorubicin alone in the first-line treatment of patients with advanced soft-tissue sarcoma.

The FDA has granted a priority review to olaratumab, in combination with doxorubicin, for the treatment of soft-tissue sarcoma.

Older soft-tissue sarcoma patients undergoing surgery derive greater benefit from radiotherapy than younger patients, according to a surprising analysis of more than 15,000 individuals.

While continuing to warn against use of laparoscopic power morcellators for the removal of uterus or uterine fibroids in most women, the FDA is allowing the marketing of a containment system for use with certain power morcellators to isolate tissue not suspected to be cancerous.

The tyrosine kinase inhibitor pazopanib appeared to have differing effects among different histologic subtypes of soft-tissue sarcoma, according to a new study conducted in Japan.

An analysis of randomized controlled trials in soft-tissue sarcoma over 40 years found that progression-free survival and response rate are reasonably well correlated with overall survival, and are thus acceptable surrogates to use.
A novel high-throughput screening assay was used to identify a number of compounds that could potentially offer therapeutic benefit in Ewing sarcoma.

A new study validated a prognostic nomogram for retroperitoneal sarcoma using a large, external cohort. The nomogram incorporates six variables, and provided strong concordance with observed disease-free and overall survival.
A phase I trial found that panobinostat combined with epirubicin is well tolerated and could offer benefit in patients with refractory sarcoma.

The FDA has granted orphan drug designation for the novel agent known as TRC105, in development by Tracon Pharmaceuticals, for the treatment of soft-tissue sarcoma.

The US Food and Drug Administration announced the approval of eribulin (Halaven) for the treatment of unresectable, metastatic liposarcoma in patients who have received prior chemotherapy with an anthracycline.