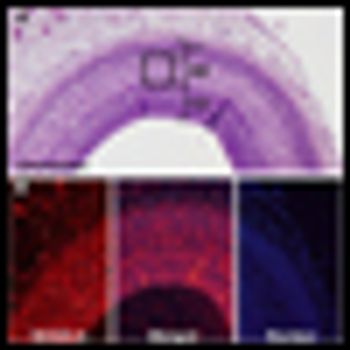
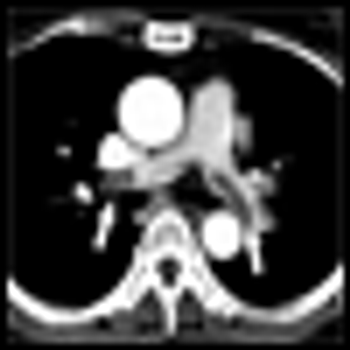
Because there has been little empirical evidence to back up patients’ stories, chemobrain has been met with skepticism by the medical community and has been a painful puzzle for many patients who couldn’t quite put a name to what they were feeling. This cognitive impairment-characterized by loss of memory and stumbling on words, among other symptoms-has come to be known as “chemobrain” or “chemofog.”