Prostate Cancer
Latest News

Latest Videos
CME Content
More News

Men diagnosed with mCPRC who are treated with 117Lu-PSMA may be assessed using nomograms to help predict outcomes.
Adjuvant radiation therapy, when compared with early salvage radiation therapy, reduced the risk of all-cause mortality for patients with adverse pathology at radical prostatectomy.
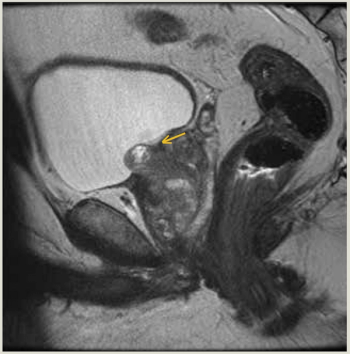
This is the case of a man, aged 56 years, who presented with urinary intermittency, frequency, urgency, and dysuria 5 months after undergoing focal laser ablation (FLA) of Gleason 3 + 4 = 7 prostate cancer (PC).

During a discussion at 2021 ASCO, Neal D. Shore, MD, said he was most excited to see data from the phase 3 VISION trial of lutetium-177-PSMA-617 in metastatic castration-resistant prostate cancer.

New research indicate that men with positive pelvic lymph nodes or a prostatectomy Gleason score of 8 to 10 with disease extending beyond the prostate may benefit most from adjuvant radiation therapy
Radioligand therapy 177Lu-PSMA-617 receives breakthrough designation from the FDA based on better overall survival outcomes in patients with metastatic castration-resistant prostate cancer.
Almost all patients with nonmetastatic castration-resistant prostate cancer treated on the phase 3 ARAMIS trial were able to receive the full planned dose of darolutamide for their disease.
Adding abiraterone acetate and prednisone to androgen-deprivation therapy and docetaxel improved radiographic progression-free survival for men with de novo metastatic castration-sensitive prostate cancer.
Despite failure to reach the primary end point of statistically significant overall survival benefit, TAK-700 plus androgen-deprivation therapy for metastatic hormone-sensitive prostate cancer may be a valid treatment option for some patients.
Disparities regarding comprehensive genomic profiling came to light during a presentation at the 2021 ASCO Annual Meeting.

For men with asymptomatic or mildly symptomatic mCRPC, using bone-protecting agents during treatment with radium-223 plus enzalutamide controlled the risk for fractures.

CancerNetwork® spoke with 2 experts to learn more about focal therapy and its role in treating localized prostate cancer moving forward.

Phase 1/2 data presented at the 2021 ASCO Annual Meeting for the novel PSMA-targeting agent HPN424 show tumor responses in patients with pretreated metastatic castration-resistant prostate cancer.

A 40% reduction in the risk of death was observed when 177Lu-PSMA-617A was added to standard of care therapy in patients with PSMA-positive metastatic castration-resistant prostate cancer.
Patients who underwent advanced molecular imaging had higher rates of disease-free survival, according to new research.

The approval of 18F-DCFPyL comes on the heels of positive results from the company-sponsored research in the CONDOR and OSPREY trials investigating the imaging agent for prostate cancer.

The ready-to-use subcutaneous formulation of leuprolide mesylate, Camcevi, was approved for use in patients with prostate cancer by the FDA.

Promising results from a phase 1b trial of cabozantinib in combination with atezolizumab for patients with high-risk, locally advanced or metastatic castration-resistant prostate cancer are expected to lead to regulatory submission.
Prostate cancer in African American men was improved with higher rates of screening for prostate-specific antigen.

The results from a trial evaluating the Genomic Prostate Score did not find an increase in active surveillance acceptance amongst a population of predominantly Black men with favorable-risk prostate cancer.
Thomas J. Polascik, MD, reflects on a manuscript written by his colleagues regarding the use of focal therapy to treat localized prostate cancer.
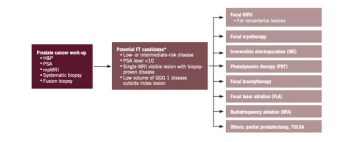
Focal therapy for prostate cancer could balance undertreatment and overtreatment of localized prostate cancer for highly selected patients. Long-term oncologic outcomes are not yet available for any modality. Patients should be informed regarding currently available outcomes, the necessity of adherence to a stringent follow-up protocol, and the possible need for additional targeted therapy or future radical treatment in case of recurrence.

A technology that uses AI to deliver focal laser therapy to prostate tumors was granted breakthrough device designation by the FDA, which will allow for the swift development and commercialization of the product.

E. David Crawford, MD, spoke about the use of transitional disease and other redefined disease states in prostate cancer, while Rana McKay, MD emphasized the need for clinical trials to validate current treatments and practices.

This article reviews new imaging technologies that may improve early detection of biochemical recurrence after definitive treatment for prostate cancer. This article also features insights from oncology experts Raoul Concepcion, MD; Judd Moul, MD; Brian Helfand, MD; Munir Ghesani, MD.