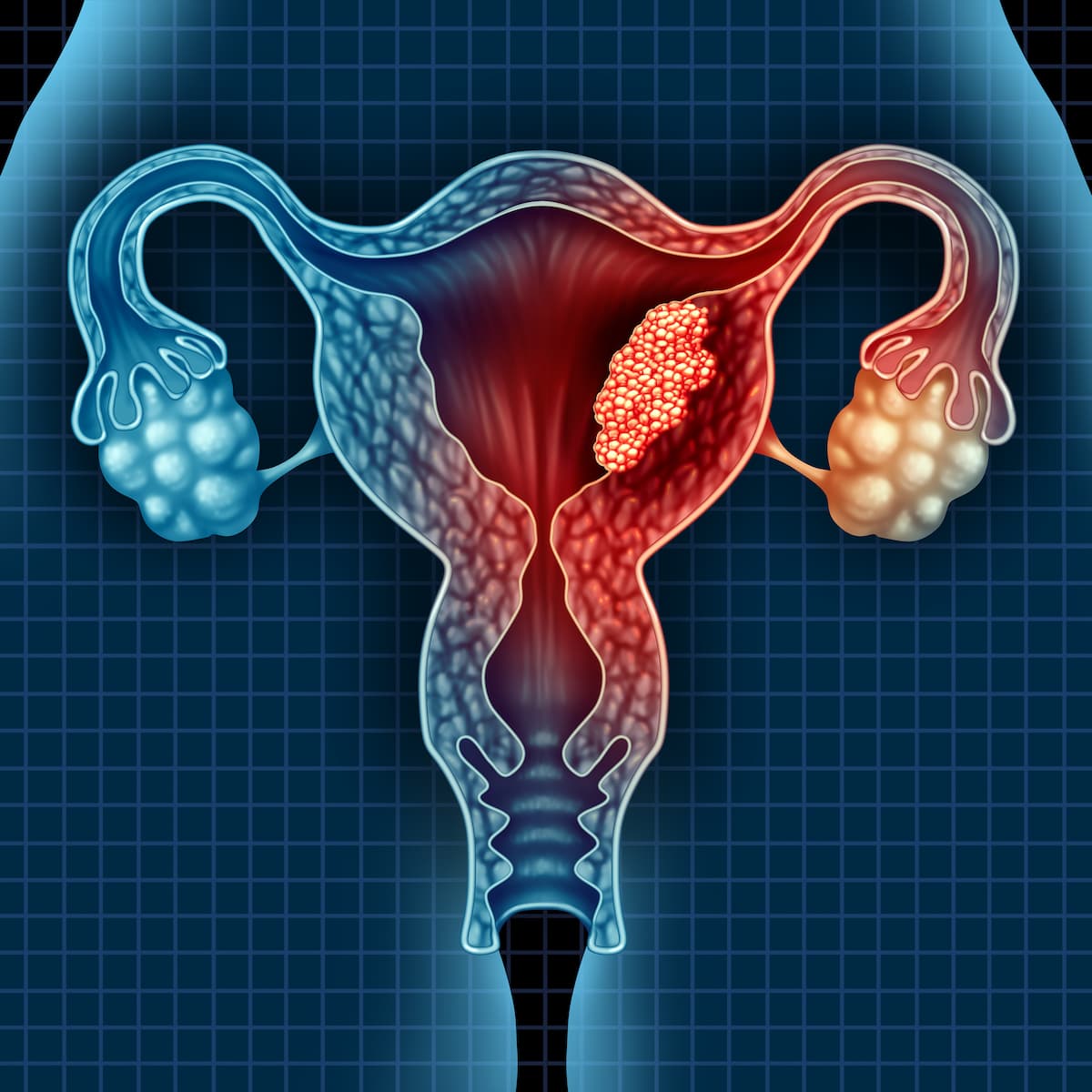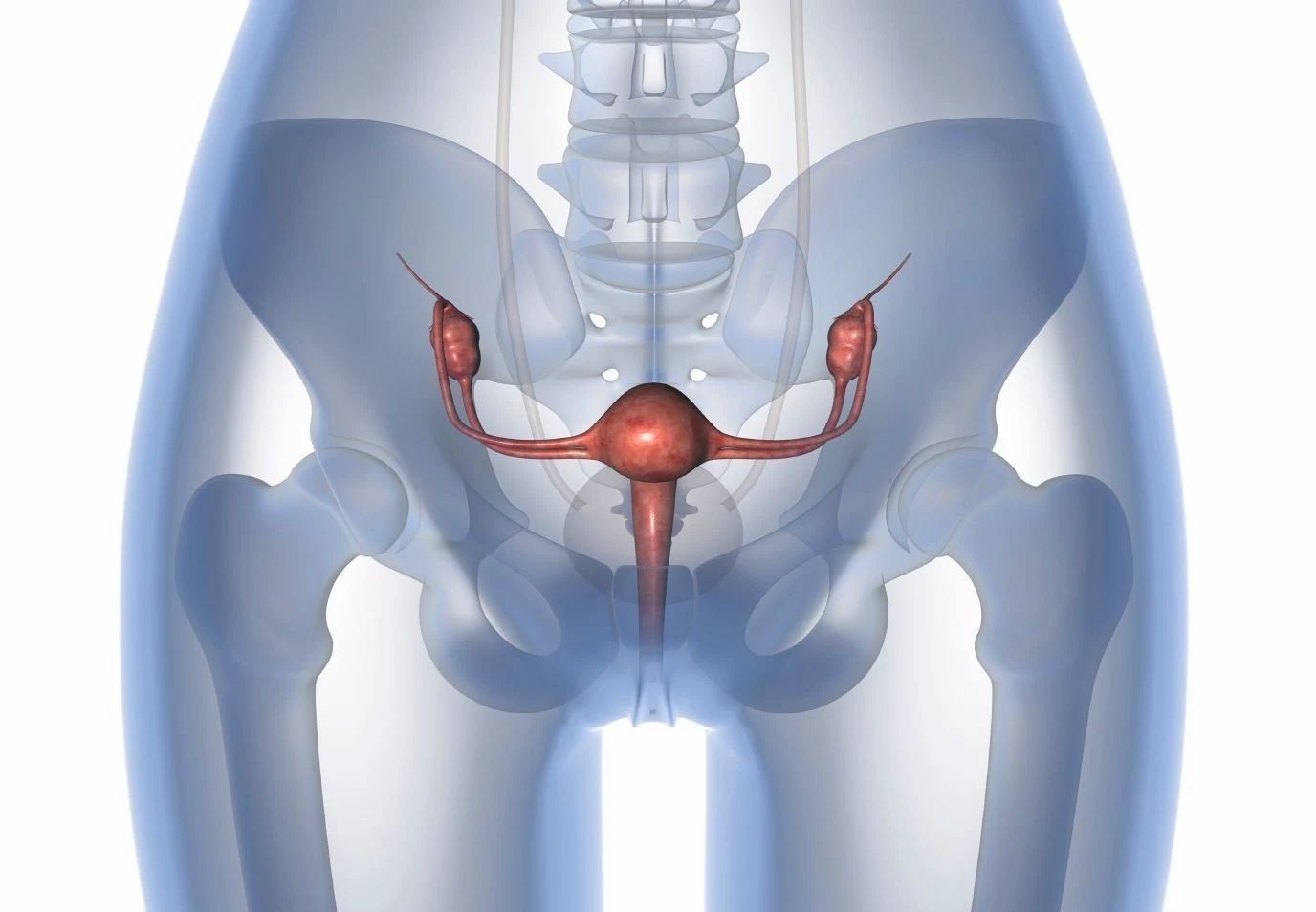
Endometrial Cancer
Latest News
Video Series

Latest Videos
Shorts




Podcasts
More News

The SISTER trial was the first national randomized trial to demonstrate significantly improved outcomes for black women with endometrial cancer.

A panel of oncologists, pharmacists, and physician assistants discussed prevalent regimens for patients with recurrent endometrial cancer.

The Promega OncoMate MSI Dx Analysis System was previously approved to identify Lynch syndrome in patients with CRC.
A significant survival improvement was observed among patients with dMMR carcinomas who received atezolizumab/chemotherapy.

The most pronounced overall and recurrence-free survival benefit with chemoradiotherapy was observed among patients with TP53-abnormal endometrial cancer.
Data from the phase 1/2 RAINFOL-01 trial support the agency’s regulatory decision on rinatabart sesutecan in advanced endometrial cancer.

Factors such as language spoken, social vulnerability index characteristics, and insurance type were found to alter endometrial cancer diagnoses and led to worse outcomes.

Patients who had recurrence in the radiation field experienced similar responses vs those with recurrence outside the radiation field.
The adverse effect profile of abemaciclib plus hormonal therapy was comparable with prior reports of CDK4/6 inhibitors.

The median PFS in patients with pMMR TP53 wild-type endometrial cancer was 39.5 months with selinexor and 4.9 months with placebo.

Puxitatug samrotecan was well tolerated in patients with advanced or metastatic endometrial cancer.
A slightly higher complete response rate was observed with the metformin regimen vs with the levonorgestrel-releasing IUD alone in endometrial cancer.
No deaths or significant adverse effects were reported in the screened population among those who received hysteroscopic resection for endometrial cancer.

Cadonilimab plus lenvatinib appeared to have a manageable safety profile in a phase 2 trial.

Adding maintenance olaparib to durvalumab/chemotherapy in pMMR endometrial cancer improved PFS among those with detectable ctDNA at baseline.

Dostarlimab/chemotherapy elicited a 31% decrease in the risk of death compared with placebo/chemotherapy in locally advanced/recurrent endometrial cancer.

Risk factors and biomarker testing in conjunction with cancer staging may better identify ideal treatment for endometrial cancer than cancer staging alone.

Deep and durable responses were observed with lenvatinib plus pembrolizumab in patients with stage III/IV recurrent endometrial carcinoma.
The addition of pembrolizumab to chemotherapy with or without radiation did not improve DFS across the intent-to-treat population of KEYNOTE-B21.

“The dMMR population, which are patients who have deficiency in their mismatch repair proteins, had the most pronounced impact in PFS, and we’re seeing that trend for prolonged periods of time; we may be curing many of these patients,” said Ritu Salani, MD.

Subgroup analysis data from DUO-E support the European approval of durvalumab-based treatment in primary or recurrent endometrial cancer.

Data from the RUBY trial support the expanded approval of dostarlimab plus chemotherapy in patients with advanced/recurrent endometrial cancer.

A statistically significant and clinically meaningful improvement in progression-free survival in the phase 3 trial DUO-E support the recommendation.

Tissue biopsy was recommended among Black patients at risk of endometrial cancer over the common ultrasonography triage strategy.

Patients with primary advanced/recurrent endometrial carcinoma may now receive pembrolizumab plus chemotherapy as indicated by the FDA.