
Data from the phase 3 EMBER-3 trial support the approval of imlunestrant in this breast cancer population.

Russ Conroy is an Associate Editor for CancerNetwork. He grew up in Hillsborough, New Jersey, and graduated from Rutgers University-New Brunswick in 2022.
On the weekends, he likes to unwind by playing video games with friends, tailgating at Rutgers football games with his family, or building his music collection with a visit to Princeton Record Exchange.

Data from the phase 3 EMBER-3 trial support the approval of imlunestrant in this breast cancer population.
The frontline treatment algorithm in SCLC is poised to look “radically different” in the upcoming years, according to Anne Chiang, MD, PhD.
Data from the phase 3 evERA trial show a trend toward improved overall survival with giredestrant plus everolimus in this breast cancer population.

Phase 1b/2 BGB-11417-105 trial data showed low rates of high-grade infection and hematologic toxicity with sonrotoclax-based therapy in multiple myeloma.

Data from the TANDEMM trial show enduring responses with concurrent anti-GPRC5D and anti-BCMA therapy in relapsed/refractory multiple myeloma.

Responses observed with the elranatamab-based regimen in the MagnetisMM-6 trial are anticipated to deepen with longer follow-up.

Data from the IDeate-Lung01 trial support the potential role that ifinatamab deruxtecan may play in the management of extensive-stage small cell lung cancer.

Findings from the 2025 World Conference on Lung Cancer reflected key updates in the management of NSCLC, SCLC, and other lung cancer types.
Subgroup data from KEYNOTE-671 support the use of perioperative pembrolizumab in stage II or III non–small cell lung cancer of any clinical nodal status.

The performance of the latest Shield algorithm underwent validation in an expanded cohort of individuals enrolled on the ECLIPSE study.

Data from the AQUILA trial support early intervention with fixed-duration subcutaneous daratumumab for those with high-risk smoldering multiple myeloma.

Patients with recurrent or metastatic cervical cancer in Hong Kong are now eligible to receive treatment with tisotumab vedotin.
Data from KEYNOTE-585 showed that adding pembrolizumab to chemotherapy did not negatively impact health-related quality of life vs placebo/chemotherapy.
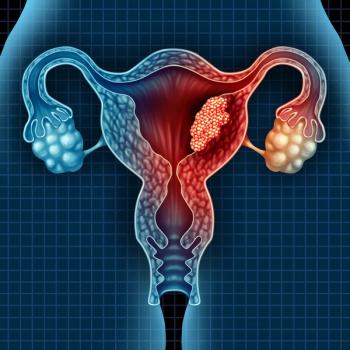
Data from the phase 1/2 RAINFOL-01 trial support the agency’s regulatory decision on rinatabart sesutecan in advanced endometrial cancer.

Developers will now initiate the phase 3 MarsLight-11 trial of IBI363 among patients with immunotherapy-resistant squamous non–small cell lung cancer.
Induction chemotherapy may allow investigators to biologically select patients with a favorable prognosis who benefit most from chemoradiotherapy.

Bridging therapy with talquetamab achieved “notable” disease control among patients with multiple myeloma in a retrospective study.

Prior data from the STRESS-LUNG-1 trial introduced emotional distress as a “psycho-biomarker” for immunotherapy efficacy in non–small cell lung cancer.

Phase 3 findings may contribute to the selection of triplet or quadruplet therapies in newly diagnosed multiple myeloma via frailty-based assessments.
Phase 1 data may support continued research of amphiphile lymph node–targeted immunotherapy in solid tumors.
Data suggest that sotorasib plus panitumumab may represent a valuable new treatment option in this KRAS G12C–mutated colorectal cancer population.

Findings from the KRYSTAL-12 trial support adagrasib as a treatment option for those with disease progression on prior chemotherapy and immunotherapy.

Geraldine O’Sullivan Coyne, MD, MRCPI, PhD, shares how a new position presents a “good opportunity” to improve community-based clinical trial access.

Additional research on novel targeted therapies may be necessary to address the unmet needs in this high-grade serous ovarian cancer population.
Findings demonstrate a need to move beyond disease-related factors to address disparities in HMA treatment patterns among those with MDS.

Phase 2b findings demonstrate improved 2-year survival outcomes with OST-HER2 compared with historical control data.

Zongertinib is now approved by the FDA for patients with nonsquamous NSCLC with HER2 TKD activating mutations.
Data from the PANOVA-3 trial may support the concomitant use of TTFields plus standard of care in solid tumors across different therapeutic settings.

Supporting data for the accelerated approval of dordaviprone come from 5 open-label trials in H3 K27M-mutant diffuse midline glioma.

The intravenous formulation of tocilizumab-anoh for CRS is expected to launch in the US on August 31, 2025.