
In this issue of ONCOLOGY, Dutcher, Mourad, and Ennis provide a current review of some newer strategies in the surgical and systemic treatment options for localized and advanced renal cell carcinoma (RCC).


In this issue of ONCOLOGY, Dutcher, Mourad, and Ennis provide a current review of some newer strategies in the surgical and systemic treatment options for localized and advanced renal cell carcinoma (RCC).

Drs. Dutcher, Mourad, and Ennis have provided an excellent review of current and potential future treatments of renal cell carcinoma (RCC); we would like to highlight some salient points from a urologic perspective.

In the current critical review we discuss these emerging trends in localized and systemic treatment as well as possible interesting combinations of the two modalities. Finally, we discuss the role of the new systemic agents in non–clear cell RCC.
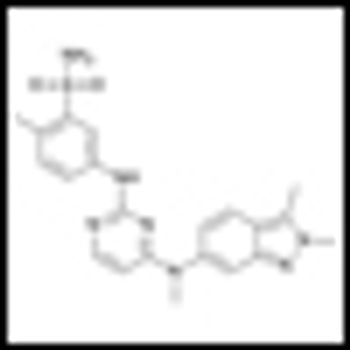
The antiangiogenic agent pazopanib demonstrated clinically meaningful activity in patients with refractory urothelial cancer in a phase II proof-of-concept study, identifying pazopanib as the first targeted compound to have clinically meaningful activity in patients with refractory urothelial cancer.

Renal cell carcinoma (RCC) had historically been regarded as a disease that was refractory to therapy once surgical options had been exhausted.

The development of vascular endothelial growth factor (VEGF) pathway inhibitors and mammalian target of rapamycin (mTOR) inhibitors for the treatment of renal cancer is a real success story.

The article by Posadas and Figlin on systemic therapy in advanced renal cell carcinoma (RCC) provides a very interesting and comprehensive review of our current knowledge concerning the treatment of RCC.

CancerNetwork speaks with Dr. Michael Atkins, who has extensive clinical experience in kidney cancer and development of various new treatments and is presenting this weekend during the renal cancer translational science session at the American Society of Clinical Oncology 2012 Genitourinary Cancers Symposium.

The US Food and Drug Administration (FDA) has approved the angiogenesis blocker axitinib (Inlyta), a twice daily oral drug, as a second-line treatment for patients with advanced renal cell carcinoma.
Researchers have identified a novel rare, germline mutation linked to a predisposition for both melanoma and renal cell carcinoma (RCC) that results in a five-fold increased risk of developing melanoma, RCC, or both.
Scientists at Jennerex, Inc. in San Francisco, and collaborators from University of Pennsylvania and the University of Ottawa in Canada have just engineered a poxvirus, JX-594, to selectively replicate in tumor cells that have an activated EGFR/ Ras pathway, but not in normal tissue.

The patient is a 43-year-old man who was initially evaluated at an outside institution for unexplained anemia and who was found to have a large right kidney mass. He underwent a radical nephrectomy for a 19-cm large-cell, poorly differentiated neoplasm, consistent with pleomorphic, epithelioid angiomyolipoma (EAML) with extensive necrosis and cytologic atypia.

The authors present an interesting case of a very rare renal neoplasm, malignant epithelioid angiomyolipoma (AML), which belongs to a family of mesenchymal tumors known as perivascular epithelioid tumors (PEComas).
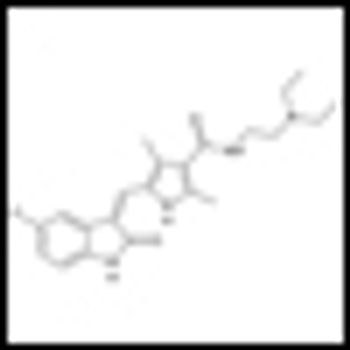
A retrospective study of metastatic renal cell carcinoma (mRCC) patients published in the journal Cancer found that patients treated with tyrosine kinase inhibitors (TKIs) had better overall survival and less-frequent metastasis to the brain.
Following a retrospective analysis which may be the largest of its kind to date, a multicenter team of investigators says treatment-related hypertension may be a useful biomarker of superior clinical outcome with sunitinib (Sutent) in patients with metastatic renal cell carcinoma (RCC).

My 2002 article provided an overview of the various interleukin-2 (IL-2)–based treatment regimens that had been explored over the preceding two decades

In 2010, we have the clinical opportunity to choose among several “targeted” agents when treating patients with metastatic renal cell carcinoma (RCC).

Newly developed targeted agents in the management of advanced renal cell carcinoma have given us treatment options in this disease not imagined a decade ago.

Listen to internationally regarded oncologist and clinical investigator, Robert J. Motzer, MD, attending physician at Memorial Sloan-Kettering Cancer Center, discuss the past, present, and future trends in renal cell carcinoma.

In this exclusive ASCO podcast, Andrew J. Armstrong, MD, ScM, Assistant Professor of Medicine and Surgery Duke Comprehensive Cancer Center, Departments of Medicine and Surgery, Divisions of Medical Oncology and Urology, addressed the vexing clinical issue of chemotherapy’s limited effectiveness in advanced renal cell carcinoma.

Expert Perspectives: Key opinion leaders in gastrointestinal cancers, renal cell carcinoma and hematological malignancies cover the best sessions and abstracts of this year’s ASCO meeting…podcasts, interviews, on-site coverage.

Patients who undergo resection of all metastatic kidney lesions, especially of disease that has spread to the lungs, appear to have outcomes as good as patients who have only a single surgery.

Approved Drugs: Pazopanib (Votrient) Indications
The patient is a 26-year-old woman with a complex oncologic history. At 1 year of age, she was diagnosed with a stage III abdominal neuroblastoma, which was treated, and again at age 9, she had a recurrence of neuroblastoma in the left axilla. She was in her usual state of good health until 18 months ago, when she presented with hematuria and was found to have a right-sided kidney mass.

Using renal mass biopsy to guide treatment decisions for small tumors is cost-effective relative to direct surgery, and can spare many patients unnecessary surgical procedures, according to Massachusetts General Hospital researchers.