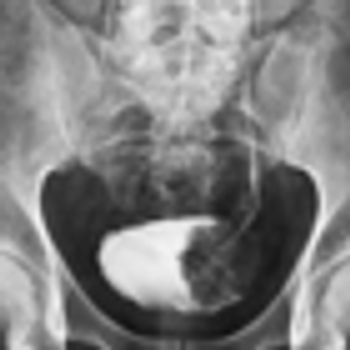
General practitioners took longer to suspect a diagnosis of bladder or renal cancer in women compared with men, according to a recent study.

General practitioners took longer to suspect a diagnosis of bladder or renal cancer in women compared with men, according to a recent study.
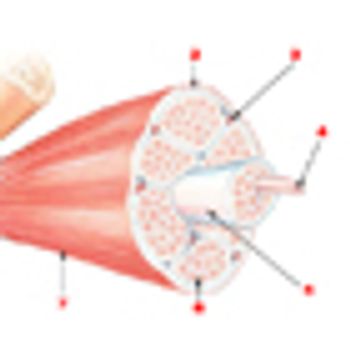
A high skeletal muscle density has been linked to a twofold prolonged survival in patients with metastatic renal cell carcinoma compared with patients with low muscle density, according to a new study.

There has never been a referendum on IL-2. True, in the era of paradigm-shifting therapies, IL-2 may be overlooked at times, but it must not be excluded from the conversation.

Ultimately, as agents in both VEGF-targeted and immunotherapy classes with lower toxicity rates are developed, questions of combination and sequence will inspire clinical investigations of strategies that, it is hoped, will maximize both the quantity and quality of life for patients with RCC. Melanoma therapy drug development continues to lead the way with regard to what is therapeutically possible with immunotherapy-and suggests that HD IL-2 continues to be relevant in today’s treatment landscape.
In this review, we examine the currently approved options available for these disease processes, including the newer agents and selected combinatorial approaches under investigation, and we attempt to identify the role of high-dose IL-2 in the context of current clinical practice.
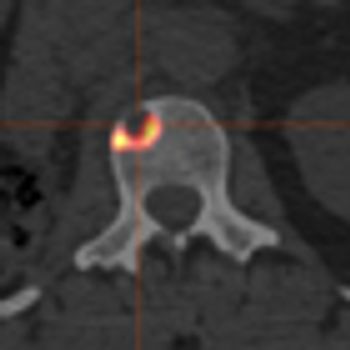
Bone metastases result in poorer outcomes for those patients with advanced renal cell carcinoma (RCC), who were treated with a molecularly targeted therapy. The results were presented in two separate analyses at the annual ASCO meeting.
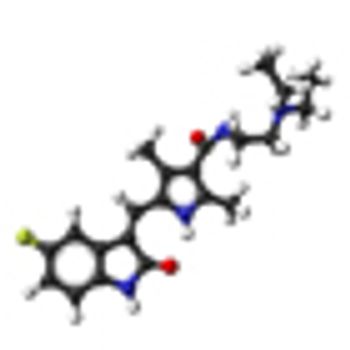
The mTOR inhibitor everolimus failed to prove progression-free survival noninferiority compared with the VEGF-targeting tyrosine kinase inhibitor sunitinib when given as first-line treatment in patients with metastatic renal cell carcinoma.
Researchers observed durable responses in patients with renal cell carcinoma treated with the PD-L1 antibody MPDL3280A. The study, which was presented at the 2013 ASCO Annual Meeting, was one of the few immune therapy trials that allowed patients with non-clear cell histologies and some clinical activity was observed in these patients.
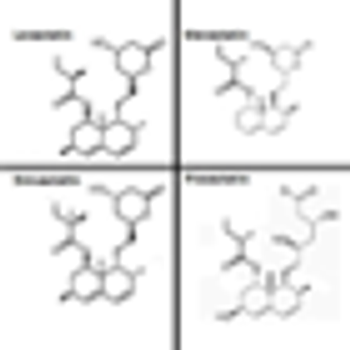
The use of statins was independently associated with improvements in overall survival and disease-specific survival among a group of patients who had undergone partial or radical nephrectomy for renal cell carcinoma.
A study found that robotic partial nephrectomy to remove kidney cancer tumors resulted in better outcomes, but also had significantly higher hospital charges. The data were presented at the annual meeting of the American Urological Association.

The FDA’s Oncologic Drugs Advisory Committee has voted 13 to 1 against AVEO's drug tivozanib for the treatment of patients with metastatic renal cell carcinoma.

A new immunoassay that tests for the presence of nicotinamide N-methyltransferase (NNMT), L-plastin (LCP1), and nonmetastatic cells 1 protein (NM23A) may be an effective method for the early detection of malignant kidney cancer.
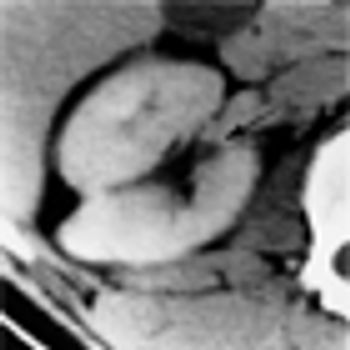
Even those renal cell carcinomas (RCCs) that are smaller than 4 cm may put patients at risk for aggressive cancer, according to a new study presented at the 28th Annual European Association of Urology Congress in Milan, Italy.

Acetaminophen and nonaspirin nonsteroidal anti-inflammatory drugs were associated with a 28% increased risk of developing kidney cancer, according to the results of a recently published meta-analysis.

High levels of physical activity were linked with a 22% decreased risk for renal cancer, according to a meta-analysis that looked at results from 19 studies that quantified the relationship with physical activity and renal cancer.

One way of framing the ethical question in this case might be: “What are my ethical obligations to provide an anticancer therapy when I think it is unlikely to benefit the patient?” The broader clinical questions involved in this case are fundamentally the same in most patients.

A trial comparing axitinib to sorafenib as first-line therapy for metastatic renal cell carcinoma showed a statistically significant improvement in progression-free survival for the drug; however, the difference failed to meet the phase III trial’s prespecified significance level of 0.025.
Results from a retrospective analysis indicate that the use of the antiangiogenic, VEGF inhibitor sorafenib for the treatment of advanced renal cell carcinoma was well tolerated in patients aged 65 years or older.
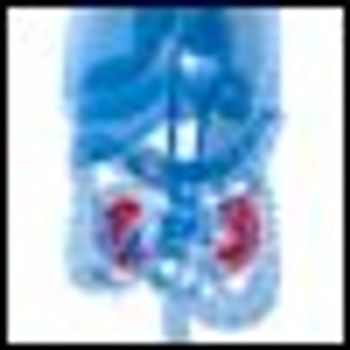
New data suggest that surveillance of patients with small kidney tumors may be a safe alternative to surgery, especially in older patients or those with comorbid conditions.
In a new study, researchers show that sunitinib, an FDA-approved TKI for advanced renal cell carcinoma, does not result in accelerated growth of kidney tumors after discontinuing treatment.
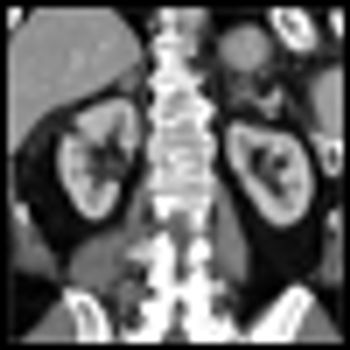
The mTOR inhibitor everolimus (Afinitor) has been found to significantly reduce the size of angiomyolipomas, the slow-growing benign tumors commonly associated with tuberous sclerosis complex (TSC) and sporadic lymphangioleiomyomatosis, according to the results of the phase III EXIST-2 study.
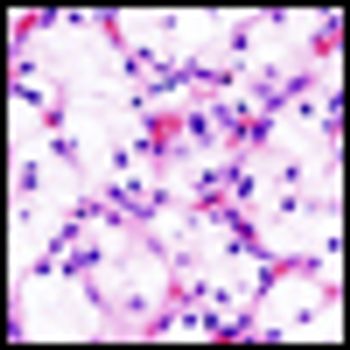
A new technique of robotic partial nephrectomy has the potential for better preservation of kidney function and better cancer control during partial nephrectomy by allowing surgeons more time to perform the procedure compared with traditional open surgery, according to the results of a recently published case series.
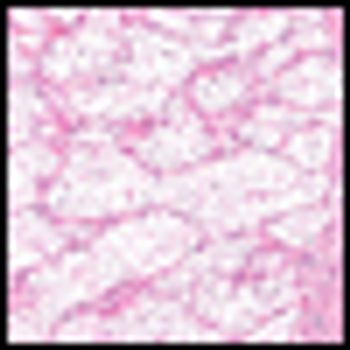
Screening patients with renal masses with iodine-124-girentuximab had both a high specificity and sensitivity for identifying clear cell renal cell carcinoma, according to the results of a newly published open-label multicenter study.
In mid-October, Pfizer announced that its phase III study being conducted examining axitinib (Inlyta) in treatment-naive patients with advanced renal cell carcinoma failed to meet its primary endpoint of improving progression-free survival compared with sorafenib.

Black patients diagnosed with renal cell carcinoma had worse survival than white patients regardless of several patient and tumor characteristics including tumor stage and size, according to data from 39,350 patients in the National Cancer Institute’s SEER program.