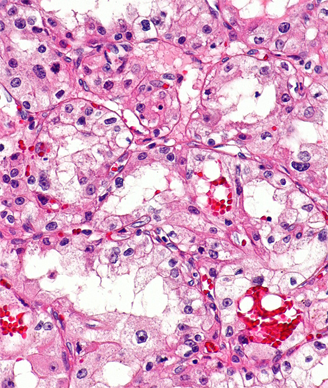
Kidney Cancer
Latest News
Latest Videos
CME Content
More News
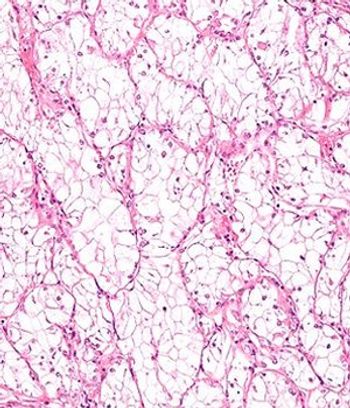
A 16-gene Recurrence Score was found to be significantly predictive of recurrence-free interval and survival in patients with stage I-III clear cell renal cell carcinoma who have undergone nephrectomy.

Patients with renal cell carcinoma who had low preoperative levels of serum cholesterol were found to have worse survival, according to the results of a recent study.

While seven drugs have been approved for clear cell renal cell carcinoma (ccRCC) since 2005, the most appropriate systemic therapy for non-clear cell renal cell carcinoma (nccRCC) is unknown.
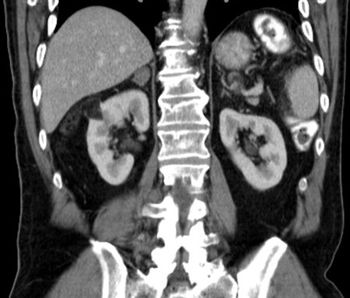
A reduced estimated glomerular filtration rate (eGFR) was found to be associated with a significantly increased risk for renal and urothelial cancer, according to the results of a recently published study.

High-dose interleukin-2 remains an important treatment for patients with metastatic renal cell carcinoma, producing durable responses even in those patients with chronic renal insufficiency, according to a recent study.

Results from two large cohorts indicated that increasing blood pressure levels were associated with an increased risk for developing kidney cancer among both men and women.
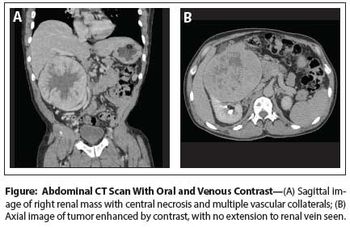
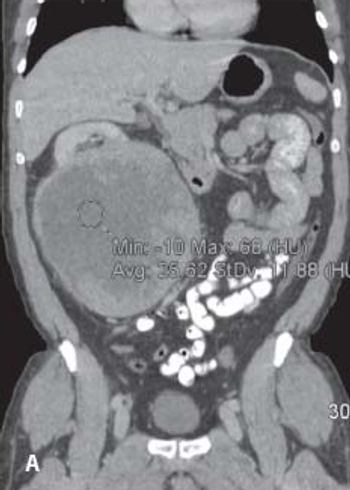
Case History: 60-year-old man with mild right side abdominal discomfort and hepatomegaly found to have large right renal mass during CT scan.
A study of patient preference revealed that patients who participated in a crossover trial of pazopanib and sunitinib for metastatic renal cell carcinoma preferred treatment with pazopanib.
Patients receiving treatment with sunitinib for advanced renal cell carcinoma experienced worse treatment-related fatigue during the first cycle of treatment with symptoms lessening after subsequent consecutive cycles.

A history of significant cigarette smoking adversely affected overall survival in patients who underwent surgery for clear-cell renal cell carcinoma, according to the results of a recent study.
The FGF inhibitor dovitinib failed to improve progression-free survival outcomes as a third-line treatment for patients with metastatic renal cell carcinoma compared with sorafenib, according to the results of a new study.

New research shows that tumors found in obese patients may be more indolent than those in nonobese patients, and this may, in part, be related to alterations in fatty acid metabolism explaining the obesity paradox in clear-cell renal cell carcinoma.
The order of the sequencing of sorafenib and sunitinib for first-line and second-line therapy did not affect progression-free and overall survival for patients with advanced renal cell carcinoma.
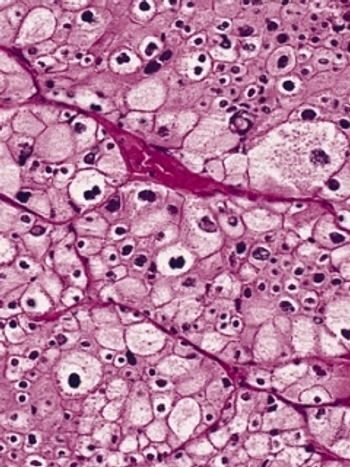
The use of angiotensin system inhibitors significantly improved the survival of patients with metastatic RCC who were on the drug for the treatment of hypertension, according to the results of a retrospective study presented at the ASCO GU Symposium.

Expressive writing is a brief and simple intervention that a recent study showed helped to reduce cancer-related symptoms and improved physical functioning in patients with renal cell carcinoma.
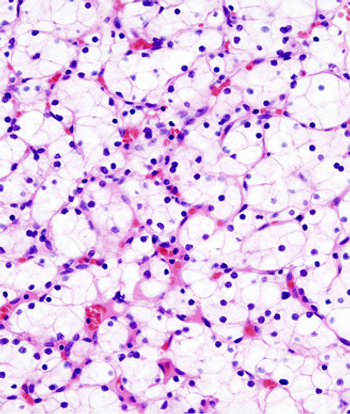
Expression of BAP1 could help identify which clear-cell renal cell carcinoma patients diagnosed with low-risk disease are the most likely to progress.
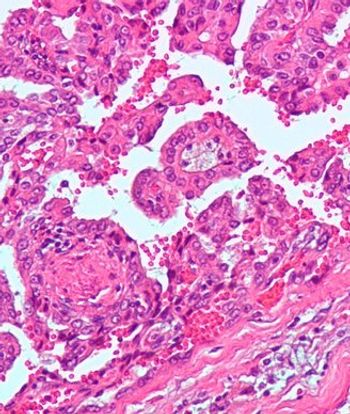
Individuals diagnosed with kidney cancer at age 46 or younger are more likely to have hereditary disease, according to a new study.
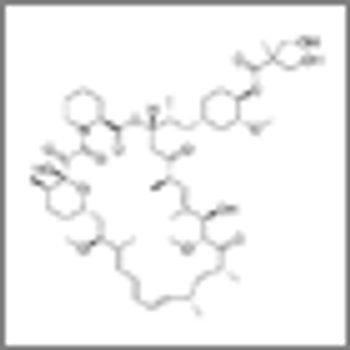
In two phase III trials, the mTOR inhibitor temsirolimus failed to improved progression-free survival outcomes for patients with metastatic renal cell carcinoma compared with other therapeutic options.
Tumor shrinkage is a valid indicator of response to VEGF inhibition among patients with metastatic renal cell carcinoma when evaluated by a single radiologist observer.

The second generation VEGFR inhibitor axitinib did not significantly improve progression-free survival in first-line treatment of patients with metastatic renal cell carcinoma compared with patients treated with sorafenib.
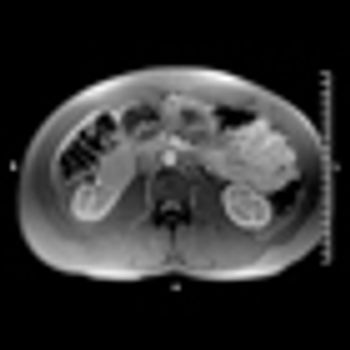
Image-guided radiofrequency ablation may be an effective alternative treatment for small renal cell carcinoma tumors, according to the results of a recent study.

Results of a recent study indicate that patient progression-free survival at 3 months and 6 months was predictive of the overall survival among metastatic RCC patients treated with interferon alpha and bevacizumab.

The mTOR inhibitor everolimus showed clinical efficacy for the initial treatment of advanced papillary renal cell carcinoma, according to the phase II results of the RAPTOR trial.

The results of the study comparing Aveo’s tivozanib to sorafenib for the treatment of metastatic renal cell carcinoma showed no improvement in overall survival, the basis for the FDA’s rejection of the company’s new drug application earlier this year.
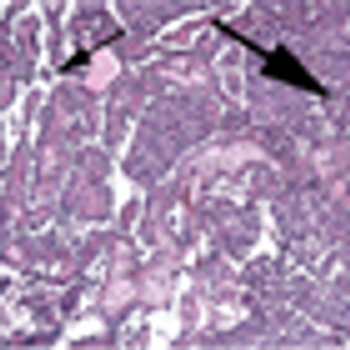
The patient is a 66-year-old male who presented to his primary care physician with a 3-week history of painless gross hematuria. He underwent a renal ultrasound that showed a left kidney mass.