Combining the EGFR inhibitors afatinib and cetuximab resulted in clinical responses among advanced lung cancer patients who had previously stopped responding to monotherapy treatment with other EGFR inhibitors.

Combining the EGFR inhibitors afatinib and cetuximab resulted in clinical responses among advanced lung cancer patients who had previously stopped responding to monotherapy treatment with other EGFR inhibitors.
Brachytherapy did not reduce rates of local recurrence after sublobar resection in patients with non–small-cell lung cancer, according to results of a new study.

A phase III trial of the insulin-like growth factor 1 receptor inhibitor figitumumab showed no benefit in survival outcomes when added to standard chemotherapy in patients with advanced non–small-cell lung cancer.

Cisplatin, etoposide, and irinotecan outperformed topotecan as second-line chemotherapy in patients with sensitive relapsed small-cell lung cancer (SCLC) in a Japanese trial, though there was substantially increased toxicity with the regimen.

The addition of ramucirumab to docetaxel improved outcomes over placebo with docetaxel as a second-line treatment of patients with advanced non-small-cell lung cancer (NSCLC), according to results of the REVEL trial.
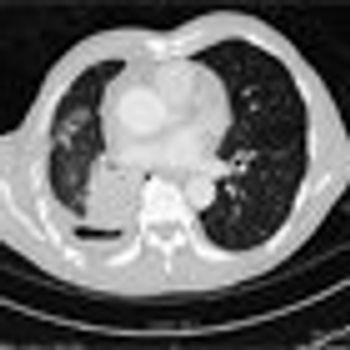
An 81-year-old Chinese male never-smoker with a medical history of hypertension and hyperlipidemia presented with a productive cough and a 5-lb weight loss over 3 months.

Several factors affect physicians’ choice of genomic tests, including availability, speed, and cost. Traditionally allelotyping have been used, since they are fast and sensitive, even though they are not comprehensive.

The addition of the anti-EGFR antibody necitumumab to gemcitabine-cisplatin chemotherapy resulted in a modest but significant benefit to patients with stage IV squamous cell non-small-cell lung cancer, according to results of the SQUIRE study presented at ASCO.

The addition of bevacizumab to erlotinib as first-line therapy yielded a significantly extended progression-free survival in patients with advanced EGFR-mutation-positive non-small-cell lung cancer (NSCLC), according to a new phase II study.

Thoracic radiotherapy along with prophylactic cranial irradiation (PCI) significantly prolonged progression-free and overall survival in patients with extensive-stage small-cell lung cancer, according to results of a new study presented at ASCO.

The addition of the MET-targeting antibody onartuzumab to erlotinib added no benefit, and may have been detrimental, to patients with MET-amplified stage IIIb/IV metastatic non–small-cell lung cancer, according to a new phase III trial.

As part of our coverage of the 2014 ASCO Annual Meeting, we discuss chemotherapy's curative role in treating lung cancer.

To ultimately find what we are actually looking for, the invasive malignant nodule in a haystack of benign lesions, new strategies and qualitative and quantitative tools are needed to propel noninvasive evaluation of solitary pulmonary nodules into the 21st century.

The number of lesions detected with low-dose CT, only some of which are early cancers, is so great that algorithms are being developed for more efficient evaluation and management of solitary pulmonary nodules. This article will discuss current tools, approaches, and concerns regarding patient care in this setting.

The simple answer, according to some, is that lung cancer screening’s time has come. However, in my opinion, the answer is not that simple.

Implementation of a national lung cancer screening program using low-dose CT will identify almost 55,000 additional lung cancer cases over 5 years, but will add $9.3 billion to Medicare expenditures.
A novel agent known as AZD9291 showed promising results in a phase I dose escalation trial among non-small-cell lung cancer (NSCLC) patients whose disease progresses after treatment with EGFR-targeting tyrosine kinase inhibitors.
Modafinil, which is commonly prescribed to manage fatigue in cancer patients, had no effect on fatigue in a new study of patients with non-small-cell lung cancer.

Earlier today the FDA granted accelerated approval to ceritinib (Zykadia) for the treatment of patients with metastatic ALK-positive non-small-cell lung cancer (NSCLC).

As part of our coverage of the AACR Annual Meeting, we discuss combination therapies and new research in the treatment of lung cancer.
New research has shown that a new assay for measuring circulating tumor DNA (ctDNA) could detect essentially all stage II-IV non-small-cell lung cancers (NSCLC).

A combination of amrubicin and cisplatin was inferior to irinotecan and cisplatin in chemotherapy-naïve patients with extensive disease small-cell lung cancer (SCLC) in a phase III trial conducted in Japan.

GlaxoSmithKline has stopped a phase III trial of a novel lung cancer immunotherapeutic agent, after determining that it may not be possible to identify which genetic subgroups may benefit from the treatment.
The new ALK inhibitor certinib showed strong antitumor activity in a phase I expansion study of patients with advanced non-small-cell lung cancer (NSCLC).

In a phase I trial, non–small-cell lung cancer (NSCLC) patients with tumors that expressed PD-L1 had significantly better outcomes with MK-3475 therapy compared with patients with PD-L1–negative tumors.