
Virginia Cancer Specialists reported they have enrolled the first patient in a new advanced lung cancer clinical trial for patients with ALK-positive locally advanced or metastatic non-small cell lung cancer.

Virginia Cancer Specialists reported they have enrolled the first patient in a new advanced lung cancer clinical trial for patients with ALK-positive locally advanced or metastatic non-small cell lung cancer.

Prescribing nivolumab only to patients with programmed cell death ligand 1-positive (PD-L1+) tumors improves its cost-effectiveness.

ALK-targeting agents such as crizotinib may work most effectively in lung cancer patients with ALK variant 1.
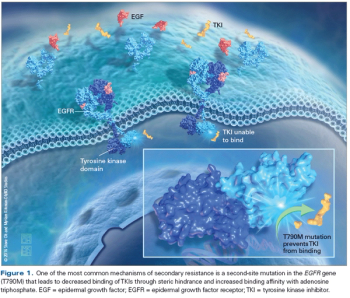
In this article, we review the available literature addressing the competing treatment strategies in EGFR-Positive Lung Cancer and attempt to clarify best treatment practices, including the emerging role of T790M-directed therapies.

Cancer cells undergo accelerated evolutionary changes; thus, under the pressure of novel generations of EGFR TKIs, we should expect to see new mechanisms of resistance.

Newly discovered microRNAs harbored by tumor-initiating cancer stem cells, drive tumorigenesis and metastasis of non-small cell lung cancer.

New steps are now being taken to revise molecular testing guidelines in an effort to improve patient selection and optimize the use of targeted therapies in lung cancer patients.

Two parts of a phase I trial found that nivolumab could be a good first-line treatment option for patients with advanced non–small-cell lung cancer.

A new combination therapy is showing promise for treating advanced solid tumors and may especially be beneficial in patients with recurrent or metastatic triple-negative breast cancer.

The FGFR inhibitor dovitinib showed modest efficacy in a phase II trial of patients with pretreated, advanced squamous cell lung cancer with FGFR1 amplification

Inflammatory arthritis and sicca syndrome appear to be immune-related adverse events among some patients undergoing cancer treatment with the immune checkpoint inhibitors nivolumab and ipilimumab.

The use of hemithoracic pleural IMRT after chemotherapy could become part of a new lung-sparing therapy in patients with malignant pleural mesothelioma.

Molecular imaging of tumor glucose metabolism might be a prognostic biomarker of NSCLC responses to the investigational immunotherapy agent atezolizumab.

An emerging field in medicine, salivary diagnostics, may be the next step in detecting treatable cancer mutations in lung cancer.

Alectinib yielded significantly prolonged progression-free survival compared with crizotinib in ALK-positive NSCLC patients, according to results of a Japanese open-label trial.

Patients with relapsed small cell lung cancer now have more options according to study results first presented at the 2016 ASCO Annual Meeting, held June 3–7, 2016, in Chicago.

An antibody-drug conjugate, rovalpituzumab tesirine, shows promising efficacy against recurrent small-cell lung cancer.

A targeted-sequencing gene panel that includes EGFR, KRAS, PIK3CA, and other mutations from circulating tumor DNA detects stage I non–small-cell lung cancer and might outperform existing tumor markers.

Matching targeted therapies to genetic abnormalities harbored by tumor types for which those therapies are not approved by the FDA might expand treatment options for some patients with advanced cancers.

The FDA has approved the cobas® EGFR Mutation Test v2, the first “liquid biopsy” blood test for detecting EGFR gene mutations in non-small cell lung cancer.

The UK Lung Cancer Screening Trial found that a low-dose CT screening program targeting high-risk individuals can find lung cancer at early stages.
British researchers have validated an IHC screening tool for more quickly and inexpensively identifying patients who should undergo crizotinib therapy for lung cancer, than is currently possible with standard FISH screening.

Using a risk-based model for CT lung cancer screening could prevent more deaths and reduce false positives compared with the USPSTF recommendations.

Afatinib resulted in improved progression-free survival compared with gefitinib in treatment-naive non–small-cell lung cancer patients with EGFR mutations.

First-line osimertinib, a targeted therapy against EGFR mutations, was found to be effective in patients with advanced non–small-cell lung cancer, resulting in a 77% overall response rate.