Endometrial Cancer
Latest News
Latest Videos

More News
Tissue biopsy was recommended among Black patients at risk of endometrial cancer over the common ultrasonography triage strategy.

Patients with primary advanced/recurrent endometrial carcinoma may now receive pembrolizumab plus chemotherapy as indicated by the FDA.

Data from the DUO-E trial support the FDA approval of durvalumab/chemotherapy for those with primary advanced or recurrent dMMR endometrial cancer.

Developers will work with investigators of the phase 3 KEYNOTE-B21 trial to share their findings with the scientific community.
The FDA has set a Prescription Drug User Fee Act date of August 23, 2024, for dostarlimab in all types of primary advanced endometrial cancer.

Although immature, overall survival data from the KEYNOTE-868 trial may support the use of pembrolizumab plus chemotherapy in patients with endometrial cancer.

Dostarlimab plus chemotherapy appears to yield favorable overall survival in patients with mismatch repair proficient endometrial cancer.

Data from the DUO-E trial support potential new durvalumab-based treatment options for patients with advanced or recurrent endometrial cancer.
Data from the phase 3 LEAP-001 study support lenvatinib plus pembrolizumab as an active combination for endometrial cancer.
SEER data showed that Black patients endometrioid endometrial carcinoma had higher 5-year cancer-related death rates compared with their White counterparts.
The survival benefit with dostarlimab/chemotherapy was deemed unprecedented in patients with microsatellite instability high primary advanced or recurrent endometrial cancer.

Results from the KEYNOTE-868 trial showed a favorable benefit of pembrolizumab plus chemotherapy across endometrial cancer subgroups.

Data from the phase 3 RUBY trial support the recommendation for dostarlimab plus chemotherapy for patients with advanced MSI-H/dMMR endometrial cancer.

Data from the phase 3 NRG-GY018 trial support the supplemental biologics license application for pembrolizumab plus chemotherapy as a treatment for those with primary advanced or recurrent endometrial cancer.

Data from part 2 of the phase 3 RUBY trial support the use of dostarlimab as a backbone for immunotherapy-based combinations in patients with primary advanced or recurrent endometrial cancer.

The European approval for dostarlimab and chemotherapy for dMMR/MSI-H, primary advanced or recurrent endometrial cancer is based on data from part 1 of the phase 3 RUBY/ENGOT-EN6/GOG3031/NSGO trial.
Total costs for endometrial cancer therapy in the first line appear to be higher for those diagnosed with metastatic disease compared with those who do not have metastatic disease.

Significant progression-free survival was seen in those with TP53 wild-type advanced/recurrent endometrial cancer who were treated with maintenance selinexor.

Investigators report a trend in overall survival improvement among patients with advanced/recurrent endometrial cancer treated with atezolizumab and chemotherapy.

The progression-free survival benefit of dostarlimab and chemotherapy extends to those with mismatch repair deficient or microsatellite instability–high advanced endometrial cancer.
Investigators report that loss of MMR mechanism is not predictive of response to chemotherapy or pembrolizumab in patients with stage III or IVA, stage IVB, or recurrent endometrial cancer.

Durvalumab plus olaparib, durvalumab monotherapy, or chemotherapy plus durvalumab saw an improvement in progression-free survival for patients with newly diagnosed advanced or recurrent endometrial cancer.
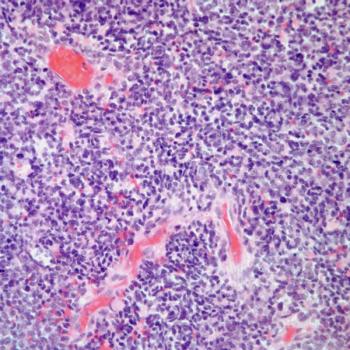
Treatment with dostarlimab plus chemotherapy followed by dostarlimab appears to improve outcomes among patients with advanced endometrial cancer and mismatch repair deficient disease in the phase 3 RUBY trial.

Data from the phase 3 RUBY/ENGOT-EN6/GOG3031/NSGO trial support the use of frontline dostarlimab plus chemotherapy for dMMR/MSI-H primary advanced or recurrent endometrial cancer.
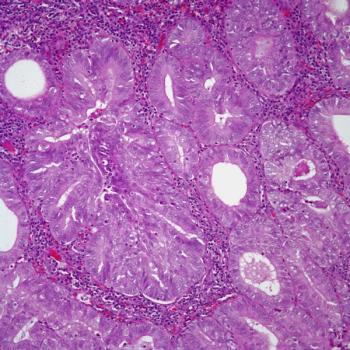
Data from the phase 3 KEYNOTE-775 trial support lenvatinib plus pembrolizumab as a standard therapy option for patients with previously treated advanced endometrial cancer.