Endometrial Cancer
Latest News
Latest Videos

CME Content
More News
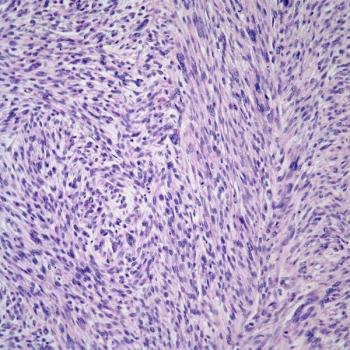
Findings from a secondary data analysis suggest a need for further research to determine risk factors that place younger patients who are Puerto Rican at a high risk of endometrial cancer.
Findings from a meta-analysis support the feasibility of administering hysteroscopic surgery plus progesterone to patients of childbearing age with various types of endometrial cancer.
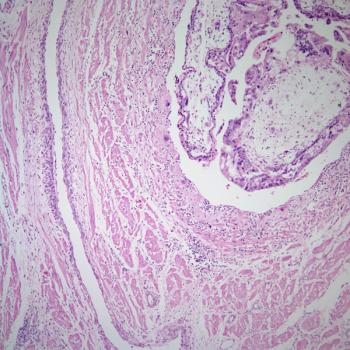
Data from a phase 1/2 trial support the fast track designation for tulmimetostat as a treatment for patients with ARID1A-mutated advanced, recurrent or metastatic endometrial cancer.
Findings suggest the presence of different genetic pathways that may drive disparities in outcomes between Black and White patients with endometrioid endometrial cancer, according to Kristin Taylor, MD.
Cutting out radiotherapy appears to be safe while improving quality of life in patients with POLE-mutated endometrial cancer.

Dostarlimab plus chemotherapy produces notable benefits among patients with advanced, mismatch repair deficient endometrial cancer in the phase 3 RUBY trial.

Findings from the phase 3 RUBY trial support the FDA’s approval of dostarlimab/chemotherapy as a treatment for advanced or recurrent endometrial cancer that is either mismatch repair deficient or microsatellite instability–high.
A progression-free survival benefit was seen in those with TP53 wild-type advanced/recurrent endometrial cancer who were treated with selinexor maintenance regardless of microsatellite instability status.

The QPOLE assay may be a fast, low-cost alternative to other next-generation sequencing tools for POLE testing among patients with endometrial cancer.
Pembrolizumab plus chemotherapy may improve clinical outcomes over placebo among patients with advanced or recurrent endometrial cancer.

Ritu Salani, MD, details the health-related quality of life benefits associated with dostarlimab in the treatment of advanced endometrial cancer, which includes improvements in back and pelvic pain.

Ritu Salani, MD, describes the concordance between blinded independent central review and provider-assessed outcomes with dostarlimab among patients with advanced recurrent endometrial cancer in the phase 3 RUBY trial.

The panel explains how low-grade serous ovarian cancer differs from high-grade, and if an immunotherapy approach is appropriate for treatment.

Robert Coleman, MD, FACOG, FACS, reviews data from a study investigating combination letrozole plus ribociclib in patients with low-grade serous ovarian cancer.
The FDA accepts a supplemental biologics license application for dostarlimab plus chemotherapy in the treatment of mismatch repair deficient/microsatellite instability-high advanced or recurrent endometrial cancer based on data from a prespecified interim analysis of the phase 3 RUBY/ENGOT-EN6/GOG3031/NSGO trial.

Health-related quality of life data support dostarlimab plus chemotherapy as a standard of care in primary advanced or recurrent endometrial cancer, according to an expert from Copenhagen University Hospital in Denmark.
Dostarlimab produces a survival benefit vs placebo in combination with standard of care therapy regardless of whether patients had mismatch repair deficient/microsatellite instability-high disease.

Drs Eskander and Lewin discuss the sequencing immune checkpoint inhibitors for women with recurrent endometrial cancer.
Data from the phase 3 DUO-E trial indicate that the safety of durvalumab/chemotherapy with or without olaparib/durvalumab or durvalumab monotherapy maintenance in recurrent endometrial cancer was consistent with previous reports of each agent.
Prexasertib is currently under investigation as part of a phase 2 trial as a treatment for patients with platinum-resistant ovarian cancer, endometrial adenocarcinoma, and urothelial cancers.

The European Medicines Agency validates the potential indication of dostarlimab/chemotherapy in mismatch repair deficient/microsatellite instability–high endometrial cancer based on the phase 3 RUBY trial.

The addition of radiation to chemotherapy did not improve overall survival in patients with advanced endometrial cancer, according to an expert from Northwestern Medicine.
Dostarlimab plus chemotherapy appears to improve progression-free survival vs placebo plus chemotherapy in patients with recurrent endometrial cancer in the phase 3 RUBY trial.

Pembrolizumab plus chemotherapy followed by maintenance pembrolizumab reduced the risk of death or disease progression in patients with mismatch repair proficient or deficient advanced endometrial cancer.
Adavosertib does not appear to be well tolerated in patients with previously treated recurrent or persistent uterine serous carcinoma in the phase 2b ADAGIO trial.