Monitoring minimal residual disease and real-time quantitative polymerase chain reaction can predict relapse in pediatric acute lymphoblastic leukemia patients.

Monitoring minimal residual disease and real-time quantitative polymerase chain reaction can predict relapse in pediatric acute lymphoblastic leukemia patients.

A phase III trial of vosaroxin failed to meet its primary overall survival endpoint in patients with first relapsed or refractory acute myeloid leukemia.

In this interview we discuss the use of newly approved agents for chronic lymphocytic leukemia, next-generation therapies in the pipeline, and how the treatment of this type of leukemia has changed.
A regimen of gemcitabine/dexamethasone/cisplatin was as effective and less toxic than the current standard of care for treating relapsed, refractory lymphoma.
Patients with non-high-risk APL can achieve better clinical outcomes when treated with ATRA plus arsenic trioxide compared with ATRA plus chemotherapy, a new study found.

Hodgkin lymphoma survivors who were treated with infradiaphragmatic radiotherapy have an increased risk of developing diabetes mellitus, says a new study.

A study of more than 4,500 patients with acute myeloid leukemia (AML) found that predicting resistance to therapy remains an elusive practice.

STAT3 inhibition using a novel compound restored sensitivity to TKIs in CML cells that had shown resistance independent of BCR-ABL1 kinase activity.
A laboratory study found that natural killer cells could be multiplied from the blood of patients to fight off precursor B-lineage acute lymphoblastic leukemia.
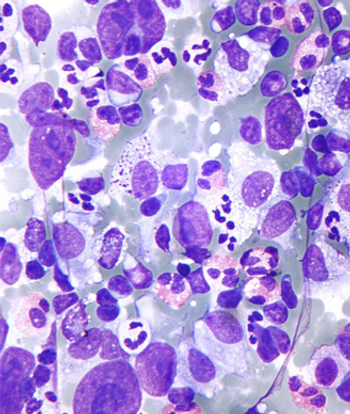
Survivors of Hodgkin lymphoma may be at increased risk of diabetes if they were exposed to radiation to the para-aortic lymph nodes and spleen during treatment.
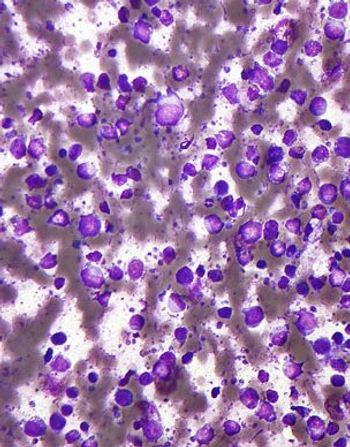
A new study confirms that patients with primary cutaneous diffuse large B-cell lymphoma who have a MYD88 mutation have a shorter disease-specific survival.
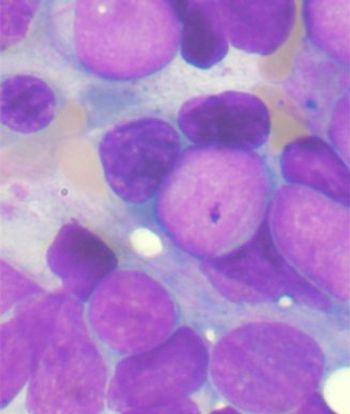
The addition of gemtuzumab-ozogamicin to standard chemo improved event-free survival in children and young adults with newly diagnosed acute myeloid leukemia.

Patients with pediatric BCR-ABL1-like acute lymphoblastic leukemia should be given risk-directed therapy based on minimal residual disease levels, according to a new study.

The FDA approved idelalisib (Zydelig) yesterday for the treatment of relapsed chronic lymphocytic leukemia (CLL), follicular B-cell non-Hodgkin lymphoma (FL), and small lymphocytic lymphoma (SLL).

Rapidly generated virus-specific T cells (VSTs) were safe and effective against five infections that commonly affect immunocompromised patients after marrow or stem cell transplantation, according to a new small study.

The FDA has granted breakthrough therapy designation to a novel treatment known as CTL019, a therapy intended for patients with relapsed/refractory acute lymphoblastic leukemia (ALL).

Is PCNSL resection safe? Evidence from clinical trials in which enrollment follows surgery-such as G-PCNSL-SG-1-is not valid proof of the safety of resection.
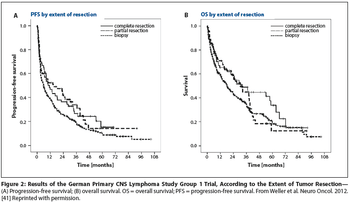
Relatively few patients with primary diffuse large B-cell lymphoma (PCNSL) will have tumors that are amenable to resection. In the absence of the highest quality data, at least it is good to know that in the modern era, patients with PCNSL are probably not harmed by judicious tumor resection.

For the practicing medical or neuro-oncologist, the treatment approach would currently not change, given that systemic therapy should be started as soon as the diagnosis of PCNSL is made and the patient is stable from a neurosurgical perspective. In most cases, one would not refer the patient back to the surgeon for additional debulking.

Lymphocyte infusions are extremely effective therapy in patients with chronic myeloid leukemia who relapse after allogeneic hematopoietic stem cell transplantation, and the timing of the infusion is relatively unimportant, according to a new study.
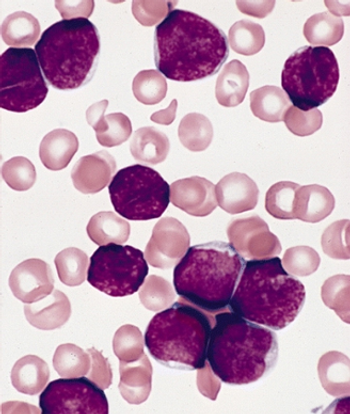
Long-term outcomes of patients treated for pediatric acute lymphoblastic leukemia (ALL) with modern treatment protocols are good, with an overall low risk for serious long-term side effects.

The FDA has approved the drug belinostat to treat patients with relapsed or refractory peripheral T-cell lymphoma.

Measuring changes in levels of BCR-ABL in patients with chronic myeloid leukemia (CML) can help predict treatment outcomes and disease progression, according to a new study.

Researchers have identified Down syndrome as a risk factor for infection-related mortality among pediatric patients with acute lymphoblastic leukemia.
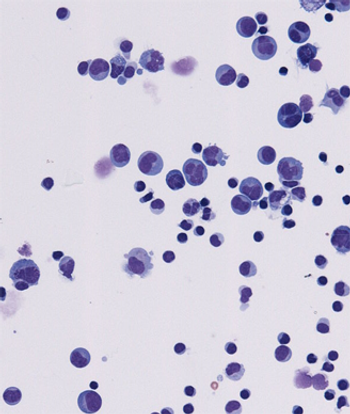
A novel inhibitor of CD19 was well tolerated and showed promising activity in patients with relapsed or refractory B-lineage non-Hodgkin lymphoma, according to a phase I study.