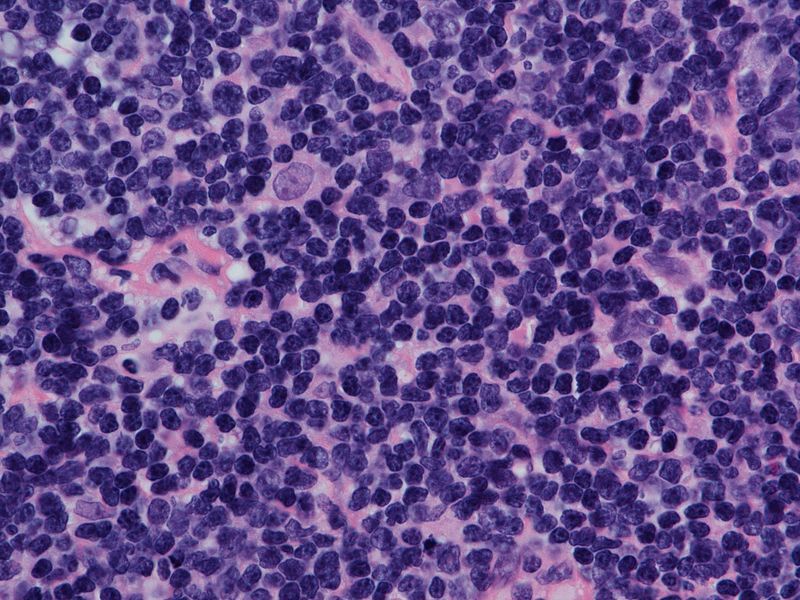
Extended follow up and preliminary findings from an extension cohort of the GO29365 study of polatuzumab vedotin (Polivy) for patients with diffuse large B-cell lymphoma confirmed earlier reports of efficacy and safety, and demonstrated continued deepening responses.