
The FDA has granted breakthrough therapy designation to a novel treatment known as CTL019, a therapy intended for patients with relapsed/refractory acute lymphoblastic leukemia (ALL).

The FDA has granted breakthrough therapy designation to a novel treatment known as CTL019, a therapy intended for patients with relapsed/refractory acute lymphoblastic leukemia (ALL).

Is PCNSL resection safe? Evidence from clinical trials in which enrollment follows surgery-such as G-PCNSL-SG-1-is not valid proof of the safety of resection.
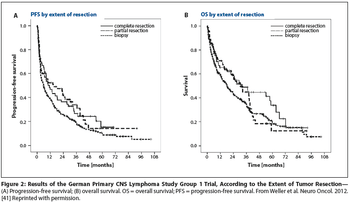
Relatively few patients with primary diffuse large B-cell lymphoma (PCNSL) will have tumors that are amenable to resection. In the absence of the highest quality data, at least it is good to know that in the modern era, patients with PCNSL are probably not harmed by judicious tumor resection.

For the practicing medical or neuro-oncologist, the treatment approach would currently not change, given that systemic therapy should be started as soon as the diagnosis of PCNSL is made and the patient is stable from a neurosurgical perspective. In most cases, one would not refer the patient back to the surgeon for additional debulking.

Lymphocyte infusions are extremely effective therapy in patients with chronic myeloid leukemia who relapse after allogeneic hematopoietic stem cell transplantation, and the timing of the infusion is relatively unimportant, according to a new study.
Long-term outcomes of patients treated for pediatric acute lymphoblastic leukemia (ALL) with modern treatment protocols are good, with an overall low risk for serious long-term side effects.

The FDA has approved the drug belinostat to treat patients with relapsed or refractory peripheral T-cell lymphoma.

Measuring changes in levels of BCR-ABL in patients with chronic myeloid leukemia (CML) can help predict treatment outcomes and disease progression, according to a new study.
Researchers have identified Down syndrome as a risk factor for infection-related mortality among pediatric patients with acute lymphoblastic leukemia.
A novel inhibitor of CD19 was well tolerated and showed promising activity in patients with relapsed or refractory B-lineage non-Hodgkin lymphoma, according to a phase I study.


A pooled analysis of three prospective trials showed that post-induction therapy PET-CT scans are highly predictive of progression-free and overall survival in follicular lymphoma patients.

As part of our coverage of the 2014 American Society of Clinical Oncology (ASCO) Annual Meeting, we discuss the pros and cons of follow-up imaging in lymphoma.

The new drug volasertib, which is in trials for the treatment of patients with acute myeloid leukemia (AML), has been granted orphan drug designation by the FDA.

Obesity or underweight status at diagnosis can influence outcomes in pediatric ALL patients, but a new study shows that the risk can be mitigated if weight status changes following treatment induction.

A novel agent known as BL-8040 will enter phase I/II testing for the treatment of chronic myeloid leukemia, according to BioLineRx, the company developing the drug.

A rare subtype of disease known as atypical chronic myeloid leukemia (aCML) has been shown to be clinically distinct from a related condition known as unclassifiable myelodysplastic/myeloproliferative neoplasm.

The FDA has approved siltuximab (Sylvant) for the treatment of multicentric Castleman disease, a disorder of the lymph nodes that is similar to lymphoma and often treated with chemotherapy and radiation.

Using both clinical and molecular markers, researchers have created a simple prognostic index for patients with cytogenetically normal acute myeloid leukemia (AML).

Our heads hit the pillow later at night as the complexity of treating DLBCL increases, but we are well rested on account of the progress that is being made.

Rationally designed clinical trials investigating novel agents in patient populations enriched for those who are most likely to benefit will be instrumental for expediting progress. With respect to DLBCL, it has become clear that one treatment no longer fits all.

With the progress in diagnostic methods that has made it possible to decipher the genetic code of DLBCL within a relatively short time, and with the increasing number of drugs that are entering clinical trials, our next big challenge is to enroll patients in trials in a timely manner.
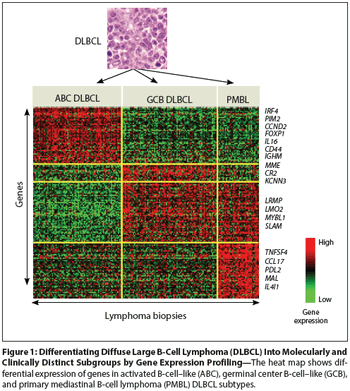
The classification of diffuse large B-cell lymphoma into three distinct molecular diseases--germinal center B-cell–like subtype, an activated B-cell–like subtype, and a primary mediastinal B-cell lymphoma subtype--has laid the foundation for the development of new agents and novel strategies that target individual subtypes.
New trial results show that a novel, oral metabolic inhibitor has demonstrated early activity in relapsed or refractory acute myeloid leukemia (AML), according to data presented at the annual meeting of the American Association of Cancer Research.

A new study of patients with diffuse large B-cell lymphoma (DLBCL) suggests that event-free status at 2 years post-treatment is a useful endpoint both for research and for patient counseling.