
Tebentafusp has been approved by the FDA based on results of the phase 3 IMCgp100-202 trial for patients harboring HLA-A*02:01 with metastatic uveal melanoma.

Tebentafusp has been approved by the FDA based on results of the phase 3 IMCgp100-202 trial for patients harboring HLA-A*02:01 with metastatic uveal melanoma.

Patients with heavily pretreated metastatic uveal melanoma appeared to benefit from treatment with a synthetic lethal combination of darovasertib and crizotinib.
The RELATIVITY-047 study highlighted a survival benefit with the combination of relatlimab plus nivolumab compared with nivolumab alone for patients with previously untreated advanced melanoma.

The combination of nivolumab plus ipilimumab demonstrated a survival benefit among patients with active melanoma and brain metastases.
Sunil A. Reddy, MD gives his perspective on the article, Metastatic Basal Cell (BCC) Arising from a Primary Cutaneous Carcinosarcoma.
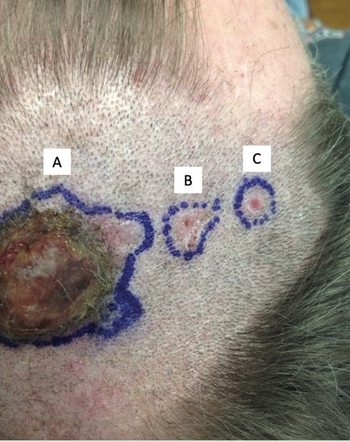
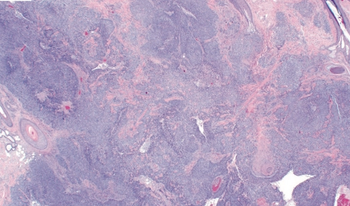
This study presents a case of a man, aged 56 years, found to have a 26-mm exophytic lesion on the vertex scalp identified to contain a distinct population of basal cell carcinoma as well as another population of spindled cells representing a poorly differentiated sarcomatous component.

FoundationOne CDx has been approved by the FDA as a companion diagnostic for patients with melanoma undergoing treatment with a BRAF inhibitor.

Pembrolizumab has been approved by the FDA as an adjuvant treatment for patients with stage IIB or IIC melanoma.
Phase 3 data indicated that the combination of nivolumab and ipilimumab followed by treatment with dabrafenib and trametinib, if necessary, resulted in greater overall survival at 2 years for patients with treatment-naïve BRAF-mutant metastatic melanoma.

Patients with melanoma, head and neck squamous cell carcinoma, and cervical cancer who had not previously received immunotherapy and were treated with lifileucel plus pembrolizumab experienced promising overall response rates compared favorably with historical data on pembrolizumab monotherapy.
Patients with melanoma who have asymptomatic brain metastases had long-lasting responses after being treated with first-line nivolumab and ipilimumab.
Sponsored by Atlantic Health System
Lifileucel, a tumor infiltrating therapy, could be the best treatment for heavily pretreated patients with advanced melanoma, with investigators at Atlantic Health System examining this treatment modality in clinical trials.
UV1 has received fast track designation from the FDA for the use in unresectable or metastatic melanoma.

John Kirkwood, MD, PhD, gives his opinion on duration of treatment for patients with melanoma.

Dr John Kirkwood explores the idea of taking drug holidays during melanoma treatment and whether it is helpful or harmful.

An expert in melanoma cancer treatment examines monitoring patients with melanoma using ctDNA.

Dr John Kirkwood discusses ctDNA as a new treatment option for patients with melanoma.

John Kirkwood, MD, PhD, addresses the idea of pseudoprogression seen in melanoma therapy.

An expert in melanoma cancer treatment relays the monitoring options for melanoma therapy and gives advice for how long these therapies should be monitored.
A retrospective review identified that clinical practice patterns have changed from 2002 to 2020, with the use of immunotherapy increasing as chemotherapy and radiation therapy use has decreased for patients with Merkel cell carcinoma.

Dr John Kirkwood discusses physician considerations that are examined before immunotherapy use in the treatment of melanoma.

John Kirkwood, MD, PhD, provides an overview of the treatment options for patients with melanoma.
Patients with previously untreated metastatic uveal melanoma who also harbored HLA-A*02:01 and were treated with tebentafusp experienced a longer overall survival compared with the control group of a phase 3 clinical trial.

A subset of patients with metastatic uveal melanoma achieved promising responses after treatment with entinostat and pembrolizumab.
Patients with relapsed/refractory unresectable or metastatic melanoma may derive benefit from alrizomadlin, which has received a fast track designation from the FDA.