
Researchers have identified several key diagnostic criteria for the diagnosis of melanoma and basal cell carcinoma with reflectance confocal microscopy.

Researchers have identified several key diagnostic criteria for the diagnosis of melanoma and basal cell carcinoma with reflectance confocal microscopy.

Researchers have shown for the first time that an oncolytic immunotherapy can have therapeutic benefit in patients with advanced melanoma.
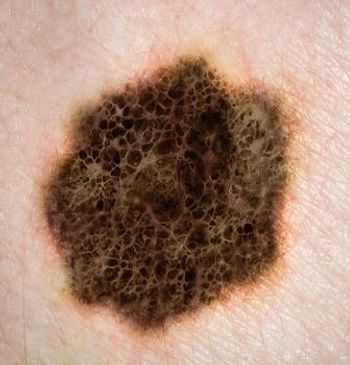
MEDI4736 appears to have an acceptable safety profile and durable clinical activity, according to a new dose-escalation study.
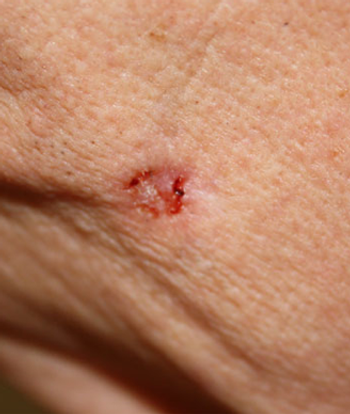
Nicotinamide, a form of vitamin B3, reduced the incidence of non-melanoma skin cancers among a group of high-risk patients, according to the ONTRAC study.
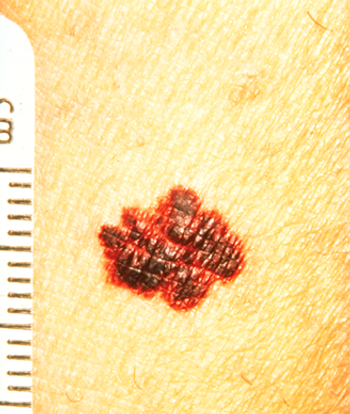
Treatment with nivolumab and ipilimumab in patients with advanced melanoma produced significantly higher rates of response and progression-free survival compared with ipilimumab alone.
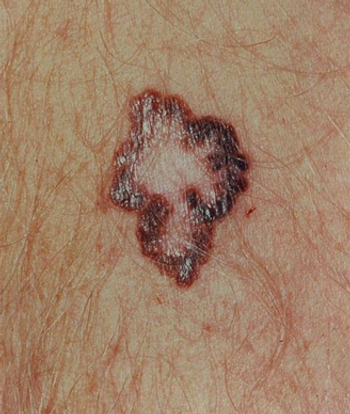
Treatment with pembrolizumab resulted in an estimated 6-month PFS rate that was almost double that of treatment with ipilimumab in advanced melanoma patients.
Advanced melanoma patients who progressed on ipilimumab were more likely to achieve an objective response when treated with nivolumab than with chemotherapy.
The measurement of levels of C-reactive protein (CRP) in the blood has been found to be an independent prognostic marker for survival in patients with melanoma.
Advanced melanoma patients who received front-line ipilimumab/dacarbazine had double the 5-year survival rate compared with those on dacarbazine alone.
A single-institution study found that patients with melanomas of 4 mm thickness who had a negative sentinel lymph node biopsy had significantly prolonged survival.
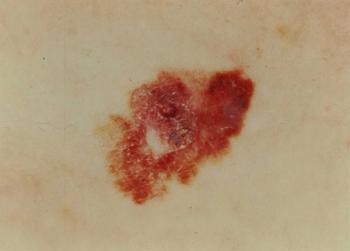
This slide show features images of melanoma, basal cell carcinoma, squamous cell carcinoma, as well as images of metastatic disease.

In early 2015, determining the optimal treatment regimen for a patient with metastatic melanoma remains challenging.

While the last several decades saw a lack of progress in treatment outcomes in this setting, the outlook has recently changed dramatically, driven by a deepening understanding of melanoma biology and host immunology.

In this article, we summarize the systemic therapies now available for melanoma, with a focus on the recently approved agents for cutaneous melanoma; discuss important considerations in selecting a treatment from the available options; and highlight some of the promising investigational approaches for this disease.

Two studies published recently showed that the use of a smartphone application was able to improve certain sun protection behaviors among study participants.

Consumption of four or more cups of caffeinated coffee per day was associated with a 20% decreased risk for malignant melanoma, according to a recent study.
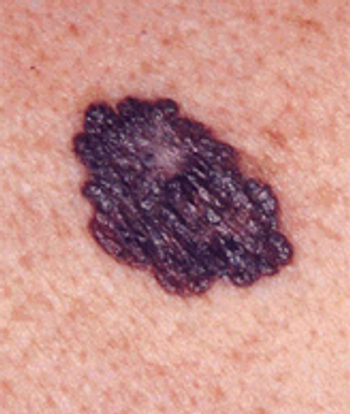
Patients with untreated metastatic melanoma had a significant improvement in overall survival when treated with a BRAF/MEK inhibitor combo.

The FDA has granted nivolumab (Opdivo) accelerated approval for the treatment of patients with unresectable or metastatic melanoma who no longer respond to other treatments.

Researchers have identified two new panRAF inhibitors that could be used to treat patients with BRAF- or NRAS-mutant melanoma, and those who have developed resistance to BRAF inhibitors.
Using whole-exome sequencing, researchers were able to define the genetic basis for deriving benefit from treatments that block CTLA-4 in melanoma.
The addition of sargramostim to the anti-CTLA-4 ipilimumab resulted in a significant improvement in overall survival in patients with metastatic melanoma.

In this interview we discuss a patient who experienced regression of BRAF-inhibitor-induced eruptive melanocytic nevi following concomitant addition of a MEK inhibitor.
Positive genetic risk information about melanoma may help to prompt people to discuss melanoma risk with a wider variety of family members, according to a new study.

A recent study has confirmed that TERT promoter mutations are common genetic mutations in cutaneous melanoma, mutations that may be linked with poor prognosis.
Treatment with a BRAF and MEK inhibitor resulted in modest clinical efficacy in patients whose melanoma had progressed after treatment with a BRAF inhibitor.