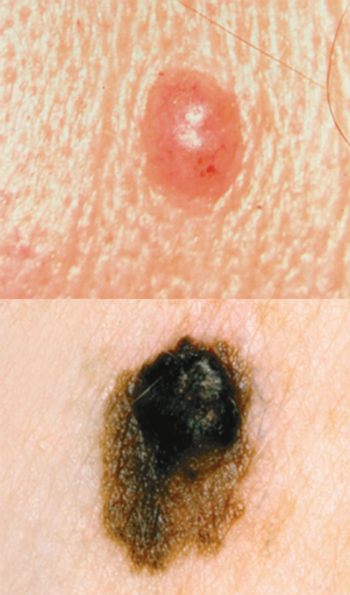
The completion of a short web-based course improved the ability of primary care physicians to diagnose and manage benign and malignant skin cancer, according to the results of a new study.
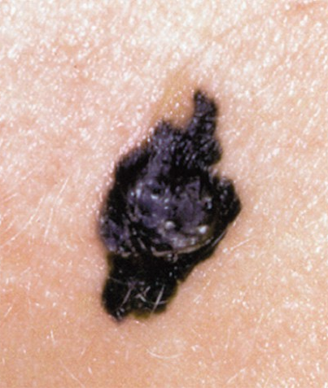

The completion of a short web-based course improved the ability of primary care physicians to diagnose and manage benign and malignant skin cancer, according to the results of a new study.

At the 10th International Congress of the Society for Melanoma Research (SMR), we spoke with Dr. Georgina Long about current targeted approaches for the treatment of advanced-stage melanoma.

Metastatic melanoma patients treated with a dabrafenib/trametinib combination experienced a 3.6-month increase in overall survival compared with patients treated with dabrafenib alone. The results were not statistically significant, partly due to the crossover design of the trial.
About one-third of patients with advanced cutaneous melanoma treated with MPDL3280A, an antibody against the programmed death 1 ligand (anti-PD-L1) showed a response in a phase I clinical trial.

The PD-1 inhibitor MK-3475 is continuing to show activity and long-term responses in patients with metastatic melanoma, according to results presented at the International Congress of the Society for Melanoma Research.
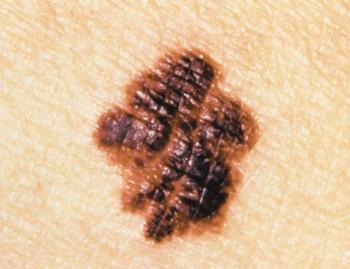
In patients with thin melanomas who are clinically node-negative, sentinel lymph node biopsy should be offered to patients with melanomas greater than 0.75 mm in Breslow thickness, according to the results of a recent study.
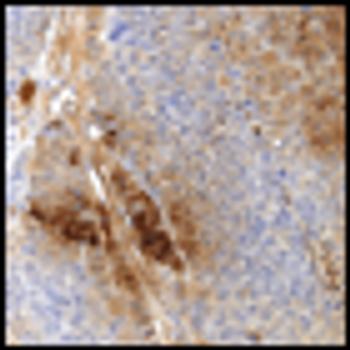
Researchers have identified a biomarker that may help physicians predict if patients with BRAF-mutant melanomas will respond to drugs designed to target BRAF.

Patients with cutaneous malignant melanoma were found to have increased their daily UV radiation dose not only while abroad or on vacation, but also on a daily basis, according to the results of a new study.

All of us treating patients with melanoma must educate our patients about the importance of participating in clinical trials with these new agents so that we can systematically validate the benefits and risks of these agents in prospectively defined patient settings.

Any minimization of therapy in the name of reducing morbidity requires careful consideration. Reducing morbidity in melanoma is certainly a laudable goal, but locoregional disease control and cure must remain our primary objectives.
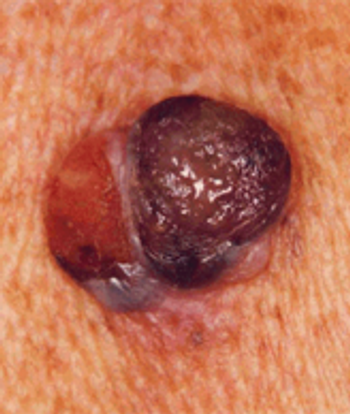
A 55-year-old woman with a history of metastatic melanoma in remission for 8 years presented to the emergency department with gross hematuria. A CT scan, ordered because the patient was in menopause, demonstrated a bladder tumor.
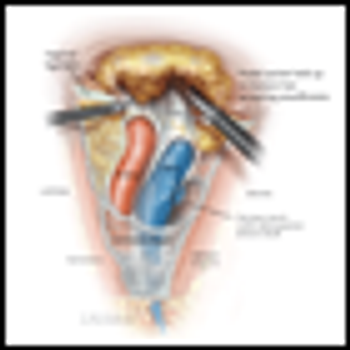
Here we review some of the most significant changes in the surgical management of melanoma that have reduced morbidity and thereby improved patient outcomes.
Patients with advanced melanoma treated with ipilimumab may be able to survive up to 10 years, according to the results of a study presented at the 2013 European Cancer Congress.

The combination of two oral targeted agents, dabrafenib (Tafinlar) and trametinib (Mekinist), has been given a priority review by the FDA for the treatment of BRAF V600-positive metastatic melanoma.

The use of GM2-KLH/QS-21 vaccination in stage II melanoma patients did not improve outcomes, according to the results of the EORTC 18961 phase III trial.

Older patients with melanoma were diagnosed with thicker melanomas, and experienced longer time to excision and a higher frequency of insufficient excision margins compared with their younger counterparts in a multicenter study in France.

A new study in early-stage melanoma patients shows that “self-eating” cells, or autophagy, can keep the cancer in check and may lead to a better prognosis.

Researchers found that circulating cell-free DNA could represent a possible predictive marker of response in advanced melanoma patients treated with dabrafenib.
Melanoma can hide in pigmented tattoos, according to a new case report describing a young man with a malignant melanoma that developed within a large tattoo.
The results of two studies indicate that combining antibodies against the programmed death 1 (PD-1) receptor with an antibody against cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4) improved treatment outcomes for patients with advanced melanoma, without a significant increase in adverse events.

There has never been a referendum on IL-2. True, in the era of paradigm-shifting therapies, IL-2 may be overlooked at times, but it must not be excluded from the conversation.

Ultimately, as agents in both VEGF-targeted and immunotherapy classes with lower toxicity rates are developed, questions of combination and sequence will inspire clinical investigations of strategies that, it is hoped, will maximize both the quantity and quality of life for patients with RCC. Melanoma therapy drug development continues to lead the way with regard to what is therapeutically possible with immunotherapy-and suggests that HD IL-2 continues to be relevant in today’s treatment landscape.
In this review, we examine the currently approved options available for these disease processes, including the newer agents and selected combinatorial approaches under investigation, and we attempt to identify the role of high-dose IL-2 in the context of current clinical practice.

Adolescent and young white men are at a more than 50% increased risk for death from melanoma than women of the same age, a new study has found. The researchers called the difference “alarming.”