
Lymphoma
Latest News
Latest Videos

CME Content
More News

Preliminary phase 2 trial data show durvalumab plus lenalidomide was superior to durvalumab alone in refractory/advanced cutaneous T-cell lymphoma.

Developing odronextamab combinations following CAR T-cell therapy failure may help elicit responses in patients with diffuse large B-cell lymphoma.

Cytokine release syndrome was primarily low or intermediate in severity, with no grade 5 instances reported among those with diffuse large B-cell lymphoma.

Here are 3 things you should know about immunotherapy in diffuse large B-cell lymphoma.

Safety results from a phase 2 trial show that most toxicities with durvalumab treatment were manageable and low or intermediate in severity.

Results from the CLOVER WaM trial saw a clinical benefit rate of 98.2% in patients with Waldenström Macroglobulinemia treated with Iopofosine I 131.

Investigators are currently evaluating mosunetuzumab in relapsed disease or comparing it with rituximab in treatment-naïve follicular lymphoma.
Phase 3 data support tafasitamab plus lenalidomide/rituximab as a potential new standard of care in patients with relapsed/refractory follicular lymphoma.

Autologous transplant did not confer significantly improved overall survival regardless of induction intensity in a phase 3 trial.
Pirtobrutinib sustained BTK inhibition and improved progression-free survival in patients with CLL and SLL, data from the phase 3 BRUIN CLL-321 trial showed.
Glofitamab with polatuzumab vedotin benefited patients with high-grade B-cell lymphomas and who failed prior CAR T-cell therapy.

Phase 2 data support epcoritamab monotherapy as a promising chemotherapy-free option for older patients with newly diagnosed LBCL.
Findings from the phase 1/2 CaDAnCe-101 trial showed that promising ORRs in R/R WM and R/R CLL/SLL stemmed from BGB-16673 treatment.

For patients with CLL and SLL, sonrotoclax with zanubrutinib yielded high rates of overall response in the phase 1/1b BGB-11417-101 study.

Tisagenlecleucel shows high rates of MRD-negative status among patients with relapsed/refractory follicular lymphoma in the ELARA trial.

Almost a 100% complete response rate was noted with Zilovertamab Vedotin/R-CHP for patients with DLBCL.
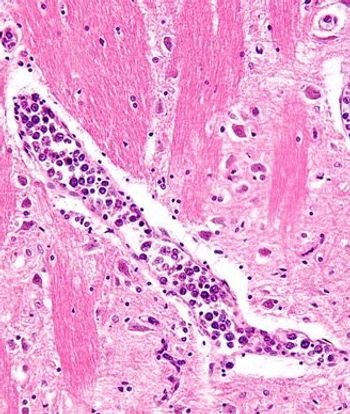
For patients with relapsed/refractory large B-cell lymphoma, PFS and OS data from CD-19-directed, 4-1BB CAR T-cell Liso-cel therapy were consistent with prior results.
For patients with relapsed or refractory diffuse large B-cell lymphoma in the US, tafasitamab elicited promising real-world efficacy outcomes.

For patients with relapsed/refractory diffuse large b-cell lymphoma, meaningful efficacy outcomes were shown with FS118.

The FDA is expected to decide on approving glofitamab in relapsed or refractory diffuse large B-cell lymphoma by July 20, 2025.

Harmonizing protocols across the health care system may bolster the feasibility of giving bispecifics to those with lymphoma in a community setting.

A leader from the adolescent and young adult lymphoma space discusses how collaboration between adult and pediatric oncologists has transformed the field.

“When thinking about treatment options for refractory DLBCL you consider: Is it safe to give an older patient CAR T-[cell therapy]?,” said Jennifer Amengual, MD.
Clinicians focused on CLL, follicular lymphoma, and MCL gathered during a Frontline Forum to discuss the use of BTKi in each respective disease state.

Establishment of an AYA Lymphoma Consortium has facilitated a process to better understand and address gaps in knowledge for this patient group.
























































