Michael L. Grossbard, MD, suggested that chemo-free regimens, including PI3K inhibitors and a more widespread use of allogeneic stem cell transplant, are being explored as treatment options in this space.


Michael L. Grossbard, MD, suggested that chemo-free regimens, including PI3K inhibitors and a more widespread use of allogeneic stem cell transplant, are being explored as treatment options in this space.
Researchers found that in patients with chronic lymphocytic leukemia receiving commercial ibrutinib, initial dose and dose modification during therapy did not appear to impact event-free survival or overall survival.
A 1-year follow-up of the phase II ZUMA-2 study found that KTE-X19 induced durable remissions in a majority of patients with relapsed or refractory mantle-cell lymphoma.

The FDA granted fast track designation to ME-401, an investigational selective oral inhibitor of PI3K delta, for the treatment of adult patients with relapsed or refractory follicular lymphoma.
Genentech recently announced their phase III study resulted in improved overall survival and composite complete remission rates for patients with previously untreated acute myeloid leukemia.
Previously unrecognized genetic structural variants in childhood leukemias could be used to evaluate the presence of minimal residual disease during chemotherapy and help to determine response to various therapies.
Hematopoietic stem cell transplantation (HSCT) with prior use of checkpoint inhibitors was found to be feasible in patients with acute myeloid leukemia and/or myelodysplastic syndromes and the use of post-HSCT cyclophosphamide as graft-versus host disease prophylaxis improves outcomes.

A study showed that adolescents and young adults with certain types of cancers saw significant improvements in their 5-year mortality rates, while other cancer types saw little to no significant improvement among the same demographic group.

The associate professor of hematology spoke with CancerNetwork® about frontline treatments available for patients with chronic lymphocytic leukemia and the toxicities present for these treatments.

The FDA granted orphan drug designation to umbralisib based on results from the phase IIb UNITY-NHL trial cohort of patients with follicular lymphoma who have received at least 2 prior lines of therapy, including an anti-CD20 monoclonal antibody and an alkylating agent.
At the 37th Annual Miami Breast Cancer Conference, Valerie Lemaine, MD, MPH, FRCSC, told physicians what they need should know and discuss with their patients about BIA-ALCL.

Kura Oncology’s leading drug candidate, tipifarnib, was granted fast track designation by the FDA to treat adults of T-cell lymphomas.

The new guidelines provide additional guidance for healthcare providers to better recognize and diagnose breast implant-associated anaplastic large cell lymphoma (BIA-ALCL).

The FDA granted priority review to a biologics license application for tafasitamab in combination with lenalidomide for the treatment of patients with relapsed or refractory diffuse large B-cell lymphoma.
This recommendation was based on an analysis of patients with diffuse large B-cell lymphoma undergoing auto-HCT in which the addition of rituximab to the BEAM conditioning regimen had no impact on transplantation outcomes.

The submission was primarily based on updated phase II efficacy and safety data for tazemetostat for patients with relapsed or refractory follicular lymphoma who have received at least 2 prior lines of systemic therapy.
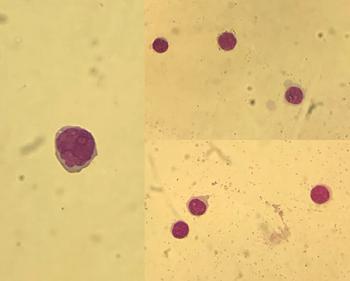
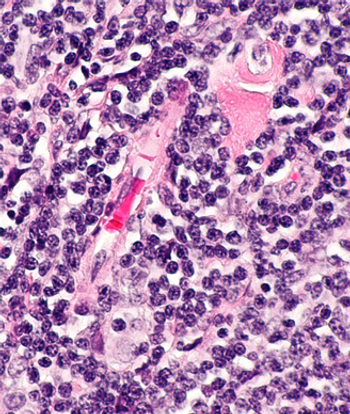
The case of a 43-year-old male with a history of B-cell acute lymphoblastic leukemia.

Bristol-Meyers Squibb Company announced that the FDA granted a priority review to its BLA for liso-cel to treat patients with relapsed or refractory large B-cell lymphoma.

The planned trial will investigate the safety and efficacy of the novel multi-tumor associated antigen T-cell therapy in patients with post-transplant acute myeloid leukemia.

Researchers found that targeting BCL-W in Burkitt lymphoma and diffuse large B-cell lymphoma may not offer wide-ranging therapeutic benefit.
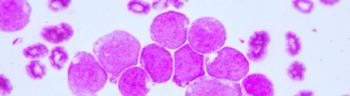
Patients with either relapsed or refractory non-Hodgkin lymphoma or chronic lymphocytic leukemia treated with CAR NK cells had a response without the development of cytokine release syndrome, neurotoxicity, or graft-versus-host disease.

Calibr received approval from the FDA to move forward with an investigational new drug to treat relapsed/refractory B-cell malignancies with a switchable CAR T-cell therapy.

Researchers indicated that a lack of understanding of the mechanism and efficacy of PD-1/PD-L1 inhibitors is the major barrier for prescription of these inhibitors in Chinese tumor treatment-related departments.
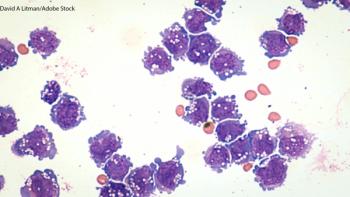
The phase I trial is evaluating cobomarsen in cancers where the disease process appears to be correlated with an increase in miR-155 levels, including adult T-cell leukemia/lymphoma, diffuse large B-cell lymphoma, and chronic lymphocytic leukemia.
Regardless of these improvements though, researchers noted that cancer survivors remain at risk for shorter lifespans, particularly when radiotherapy was utilized as a part of their childhood cancer treatment.