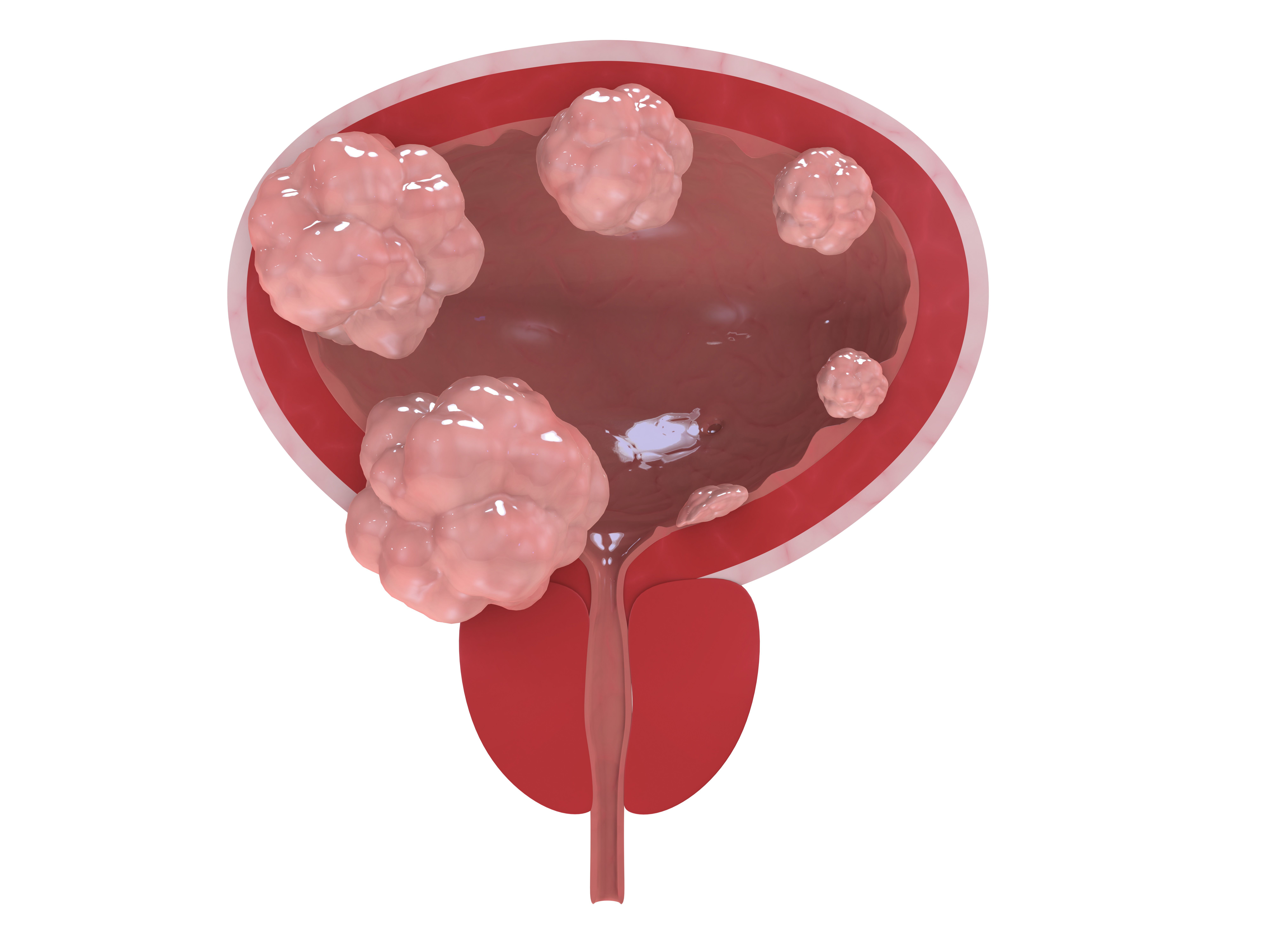
Bladder Cancer
Latest News
Latest Videos

More News

Thomas Powles, MBBS, MRCP, MD, presented results from the NIAGRA trial assessing perioperative durvalumab in patients with cisplatin-eligible MIBC.
Durvalumab improved efficacy in patients with muscle-invasive bladder cancer.

UGN-102 also demonstrated a 79.6% complete response rate at 3 months in the phase 3 ENVISION trial.

Patient-reported outcomes from the EV-302 trial found consistent efficacy and safety outcomes for patients with urothelial carcinoma.

Limited efficacy was observed when sacituzumab govitecan was given after enfortumab vedodin in metastatic urothelial carcinoma.

Adjuvant sacituzumab govitecan demonstrated a complete response, but its future in bladder cancer is uncertain after a recent protocol amendment.
Neoadjuvant avelumab plus cisplatin yielded high event-free survival in muscle-invasive urothelial carcinoma.
The CheckMate -901 trial led to the approval of the nivolumab combination for unresectable or metastatic urothelial carcinoma in the European Union.

Nivolumab plus ipilimumab has shown a response benefit when given at an increased dose for those with metastatic urothelial carcinoma.
Kirollos S. Hanna, PharmD, BCPS, BCOP, FACCC, gives his perspective on the approval of enfortumab vedotin plus pembrolizumab for patients with urothelial carcinoma.

Phase 2 data support the potential of replacing cisplatin with nab-paclitaxel as a backbone therapy for those with muscle-invasive bladder cancer.

Findings from the CheckMate-901 study support the use of frontline nivolumab plus gemcitabine/cisplatin in advanced urothelial carcinoma.

All patients with a response to TAR-200 did not have progression to muscle-invasive bladder cancer or metastatic disease in the SunRISe-1 trial.

Treatment with cretostimogene grenadenorepvec appears tolerable among patients with high-risk BCG–unresponsive NMIBC in the phase 3 BOND-003 trial.

Findings from the CheckMate-901 trial support the CHMP’s positive opinion of nivolumab plus chemotherapy in metastatic urothelial carcinoma.

Nogapendekin alfa-inbakicept is now approved for the treatment of patients with BCG-unresponsive non-muscle invasive bladder cancer.
Patients with muscle-invasive bladder cancer who remain molecular residual disease negative following cystectomy may be spared from adjuvant therapy.

Enfortumab vedotin plus pembrolizumab appears to reduce the risk of disease progression or death vs chemotherapy in locally advanced or metastatic urothelial carcinoma.
Oncology pharmacists are important to the oncology care team, as they can bring a unique perspective to treatment planning, according to Kirollos S. Hanna, PharmD, BCPS, BCOP, FACC.

Neuropathy is an adverse effect that can occur when enfortumab veodtin is administered to patients with locally advanced or metastatic urothelial carcinoma.

Data from the phase 3 EV-302 trial support an application for enfortumab vedotin plus pembrolizumab as a treatment for patients with advanced bladder cancer in China.

The Oncology Brothers recap the discussion on treatment practices for patients with bladder cancer.

Focusing on the metastatic setting, the expert panel discusses advanced kidney cancer treatment practices, highlighting recent data that are informing treatment decisions.

Karine Tawagi, MD, and Sia Daneshmand, MD, offer comprehensive insights on treatment practices for patients with muscle-invasive bladder cancer.

Sia Daneshmand, MD, and Karine Tawagi, MD, join Rahul Gosain, MD, and Rohit Gosain, MD, to discuss the treatment algorithm for non–muscle-invasive bladder cancer.