
Prophylaxis with tacrolimus/sirolimus provided equivalent graft-vs-host disease-free survival in patients undergoing matched, related donor hematopoietic cell transplantation.

Prophylaxis with tacrolimus/sirolimus provided equivalent graft-vs-host disease-free survival in patients undergoing matched, related donor hematopoietic cell transplantation.

Lymphocyte infusions are extremely effective therapy in patients with chronic myeloid leukemia who relapse after allogeneic hematopoietic stem cell transplantation, and the timing of the infusion is relatively unimportant, according to a new study.

Researchers from Japan believe they have identified a novel staging system based on hemoglobin and plasmacytoma that may help to stratify patients with multiple myeloma who are treated with novel therapeutics.
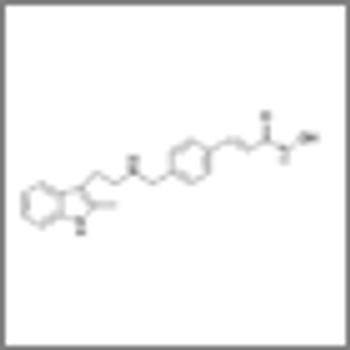
Carfilzomib given at a higher dose as a slow intravenous infusion over 30 minutes resulted in a very high response rate in a heavily pretreated myeloma population.
Long-term outcomes of patients treated for pediatric acute lymphoblastic leukemia (ALL) with modern treatment protocols are good, with an overall low risk for serious long-term side effects.

The FDA has approved the drug belinostat to treat patients with relapsed or refractory peripheral T-cell lymphoma.

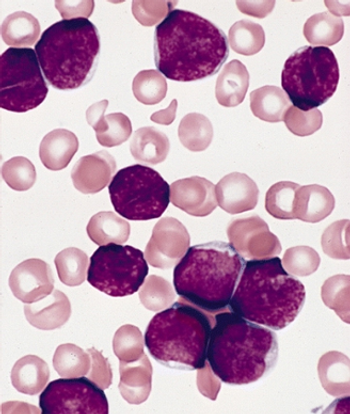
Measuring changes in levels of BCR-ABL in patients with chronic myeloid leukemia (CML) can help predict treatment outcomes and disease progression, according to a new study.
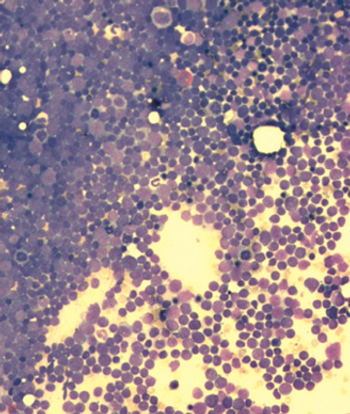
Researchers have identified Down syndrome as a risk factor for infection-related mortality among pediatric patients with acute lymphoblastic leukemia.


A comparison of melphalan, prednisone plus either thalidomide or lenalidomide found that patients assigned to lenalidomide had fewer grade 3 or higher toxicities and a better quality of life at the end of induction therapy.

This review will focus on newer FDA-approved targeted therapies associated with type II chemotherapy-related cardiac dysfunction, or generally reversible cardiotoxicity, and will provide the latest information on the incidence and clinical spectrum of cardiotoxicity associated with each therapy, modifiable risk factors where known, and the mechanisms of cardiotoxicity.
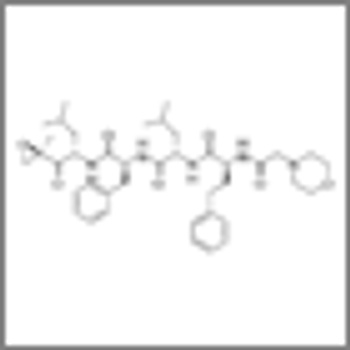
Treatment of relapsed or refractory multiple myeloma with the three-drug combination of panobinostat/bortezomib/dexamethasone resulted in a nearly 4-month improvement in progression-free survival compared to treatment with bortezomib/dexamethasone alone.
Ruxolitinib was significantly better than the best available therapy in a phase III, open-label trial of patients with polycythemia vera who were resistant to or intolerant of hydroxyurea.
A phase II trial of the selective spleen tyrosine kinase (Syk) inhibitor GS-9973 showed substantial biologic activity in patients with chronic lymphocytic leukemia (CLL).
Ibrutinib substantially increased progression-free survival and overall survival over ofatumumab in patients with chronic lymphocytic leukemia (CLL) and small lymphocytic lymphoma (SLL), according to the results of the RESONATE trial.
Treatment with lenalidomide and dexamethasone improved quality-of-life measures in multiple myeloma patients, according to a study presented at the 2014 ASCO Annual Meeting.
Continuous therapy produces drastically better progression-free survival and overall survival outcomes in patients with multiple myeloma. A novel endpoint involving time to second progression is an important tool in evaluating this type of treatment in the future.

As part of our coverage of the 2014 American Society of Clinical Oncology (ASCO) Annual Meeting, we discuss the pros and cons of follow-up imaging in lymphoma.

The new drug volasertib, which is in trials for the treatment of patients with acute myeloid leukemia (AML), has been granted orphan drug designation by the FDA.

Obesity or underweight status at diagnosis can influence outcomes in pediatric ALL patients, but a new study shows that the risk can be mitigated if weight status changes following treatment induction.

A novel agent known as BL-8040 will enter phase I/II testing for the treatment of chronic myeloid leukemia, according to BioLineRx, the company developing the drug.

A phase II trial of propylene glycol-free melphalan in patients with multiple myeloma has met its primary endpoint, according to a statement released by the drug manufacturer.

The NICE announced that newly diagnosed multiple myeloma patients will be guaranteed access to treatment with the proteasome inhibitor bortezomib (Velcade).

A rare subtype of disease known as atypical chronic myeloid leukemia (aCML) has been shown to be clinically distinct from a related condition known as unclassifiable myelodysplastic/myeloproliferative neoplasm.

The FDA has approved siltuximab (Sylvant) for the treatment of multicentric Castleman disease, a disorder of the lymph nodes that is similar to lymphoma and often treated with chemotherapy and radiation.